Study site selection
Our study was carried out on 65 plots (Fig. 2). Each plot was a 5 × 5 km square in the Transverse Mercator Projection (TMP). For the selection of 60 plots, we divided the whole territory of Latvia into 10 similar-sized TMP sectors, and randomly selected 5–7 plots in each, proportionally to the size of terrestrial part in a given sector. We also added five more randomly selected plots within the range of B.bombina, an EU Habitat Directive Annex II species, along the southern and south-eastern border of the country, because all the other random plots were outside the range of this species. Hence, the survey was semi-representative for the whole country.
In each plot we randomly selected waterbodies from orthophoto maps proportionally to (i) their presence in open versus closed landcover type, (ii) spatial distribution in four equal parts of a plot (determined as four quadrants over plot along N–S and E–W axes), and (iii) their numbers in three size categories – small (less than 0.5 ha in surface area), medium-size (0.5–10.0 ha), and large (more than 10.0 ha). We surveyed 15–35 (mean 18.6 ± SD 4.5) waterbodies and 5–10 points (6.0 ± 1.6) near larger ditches (i.e. channels for soil drainage and water flow; excludes channelled river or stream stretches with flowing water) per plot. Most of the waterbodies (91%) present on plots were small; we sampled 27% of them, and also 49% of all the medium-size, and 57% of all the large waterbodies. For waterbody dimension measurements we used orthophoto maps in the Google Earth Pro program (Google LLC, Mountain View, California, U.S.).
Field surveys
To cover peak calling activities of all anuran taxa, we visited each plot at least once during the each of the following periods: (i) April 1–15, (ii) April 16–30, and (iii) May 20–June 10. We selected days with warmest weather forecasts within given periods, when we expected high calling activity20,39,40. If we observed low activity (a calling index3 value of 1) or only few sites with call records, we repeated the survey at the given plot some days later or in the same calendar period in the following year. Surveys were not confined to certain times of day or night, except for the surveys in six plots in the range of H.arborea (a species complex 41) in south-western Latvia, which were visited only after the sunset, because this taxon typically does not vocalise in daylight23,24. We conducted a total of 298 calling surveys in 2016-2018, on average 4.6 ± SD 1.1 (range 3–7) surveys per plot.
Typically, only one observer per survey was present due to the large amount of fieldwork and the limited numbers of trained observers. During surveys the observer recorded the number of calling males at locations near waterbodies or ditches over a period of at least 5 minutes (no maximum time limit). After this, whenever it was feasible due to the condition of waterbody banks and the time of the day, the observer approached and walked around or along the waterbody or ditch for a better counting of calling males, and for counting visible breeding adults and egg clumps or strings. In some cases, with chorusing frogs, when counts were hindered by overlapping calls or banks were inaccessible and did not allow approaches close enough to distinguish individual calls, the number of calling males was recorded as an average between the minimum and maximum estimates by the observer. Observers also recorded the time of day, air temperature, wind speed (Beaufort scale), and sky conditions (clear, overcast, partial clouds) during the onset, middle and end of the survey, to confirm the overall suitability of the survey conditions.
Data processing and statistics
We used the largest number of simultaneously counted males per visit in a given site (waterbody or ditch) for population density estimations. Data from multiple years were not pooled; if we had data from several years for the same plot, we used counts only from the year with the best record, because we observed whole choruses to move between neighbouring waterbodies in consecutive years. For plots with data from several years, we estimated inter-year fidelity (IYF) in the use of breeding waterbodies. This was calculated as a ratio of waterbodies with a call record in the most productive year to waterbodies with a call record in pooled data for all years. IYF included both, true breeding site shifts and false shifts from undetected waterbodies, which we did not discriminate in our estimation.
There were many cases when the most productive survey in terms of total counted males was the most productive only for the part of the individual sites of a given plot. We estimated the contribution from a most productive survey (CMPS) as a ratio of the most productive survey score to the total calling male score in a given year. Lower CMPS values would indicate the desirability for more surveys per breeding period, and high CMPS values that fewer surveys are sufficient for data collection.
We also calculated the probability of detecting a breeding waterbody from the audial surveys (WDP) as a ratio of waterbodies with calling male records to waterbodies with any evidence of the presence of adults during the breeding period (including both, audial records and direct (adults seen) or indirect (presence of spawn, tadpoles, metamorphs) visual evidence) for a given taxon in a given year.
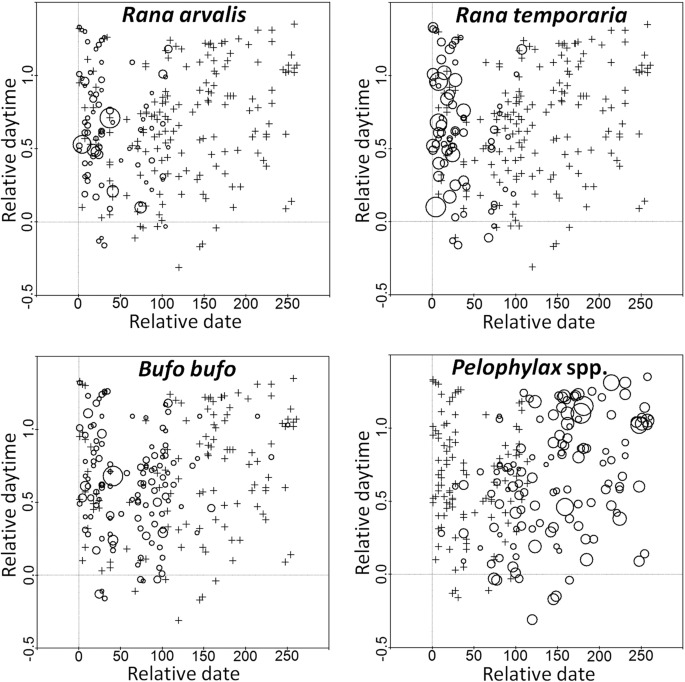
In activity analyses, we transformed calendar dates into decimal numbers. In order to remove inter-year variation in breeding season start dates, we further transformed acquired decimals into relative dates after the earliest audial record of any taxon in a given year. We also transformed time of day on the middle of the survey into decimal numbers and then into relative daytime, where 0 was the astronomic noon and 1 was the sunset time on a given date, taken from World Weather Online web-site (worldweatheronline.com, accessed 05.01.2019.). This was done to compensate for the increase in day length during the sampling period.
Our data did not fit normal distribution, so we used Poisson Regression Analysis to test statistical significance of relationships between dependent variables, such as calling males per waterbody, Dc or MAFD and various quantitative factors from our data sets. Data was transformed into counts by multiplying them by factor of 100 and rounding to integers. Our date and daytime effect analyses were done separately for each year, and here we used only data from surveys with records of a given taxon. Other relationships we studied in pooled data sets from all years. We performed all statistical analyses using Statgraphics Plus 5.0 (Statgraphics Technologies, Inc., The Plains, Virginia, U.S.) and SPSS PASW Statistics 18 (IBM Corporation, Armonk, New York, U.S.). XY(Z) charts or ‘bubble plots’ were prepared in CANOCO for Windows 4.5 software (Biometris –Plant Research International, Wageningen, The Netherlands).
Calculation of metrics
We introduced two new metrics (Dc and MAFD) to quantify frog populations. The density of calling males (Dc) is basically a waterbody category-specific extrapolation of recorded calling frogs to all the plot waterbodies, and the summation of results from all the categories given per area unit. In each waterbody category we used the total calling male score and the ratio of total to surveyed habitat in a plot. Small waterbodies (<0.5 ha) showed no correlation between their size and the number of calling males of any taxa (Generalised Linear Models, P > 0.1 in all cases), were fully within the hearing range of an observer and therefore could be treated as units in our ratio estimations. In larger waterbodies in the study area frogs typically breed only in the shallow shoreline zone (unpublished data), so there may be a correlation between the length of a shoreline and population size. Often only part of a medium-size or large waterbody is within the hearing range of an observer so in our ratio estimations we treated them as linear shoreline habitat.
The Dc metric was calculated as follows (Eq. 1):
$$Dc=frac{{sum }_{i}frac{Cast Pt}{Ps}}{S}$$
(1)
where Dc– is the density of calling males; Ʃ – represents the summation of the calculations for four waterbody categories (i): small (<0.5 ha), medium (0.5–10.0 ha), large (>10.0 ha) waterbodies, and ditches; C– is the number of calling males scored in a waterbody category i; Pt – total habitats in a waterbody category i, which is the number of all small waterbodies on the plot, or total length of waterbody perimeters for medium or large waterbodies, or length of all ditches; Ps – surveyed habitats in a waterbody category i, which is the number of sampled small waterbodies, the total length of the perimeters sampled for medium or large waterbodies, or total sampled ditch length; S – is the area of the plot.
The sampled length of waterbody perimeter or ditch was the length within the hearing range of an observer. The distance from which it is possible to hear a call depends on taxon, weather, habitat, obstacles and background noise22. Unless count results indicated larger distances, in calculations we used approximate minimum hearing distances of 500 m for Pelophylax spp. and H.arborea, 150 m for B.bufo, 100 m for R.arvalis and B.bombina, and 50 m for R.temporaria and P.fuscus. Such a distances were approximations of average values estimated from first-year surveys using GPS coordinates of calling sites, most distant points, where calls were audible and distance measurements by Google Earth Pro.
The second metric (MAFD) transfers scores of calling males into a proxy for the true adult frog population. It relies on the probability of detecting males with reference to their breeding behaviour patterns (presence in breeding waterbody and probability of being heard) to estimate their true population and sex ratios to transfer male population estimates into total adult population. Since this metric is based on maximum detection probabilities with perfect survey timing and environmental conditions (which are unlikely to be met in a random survey) the estimate would typically be smaller than the true population so we obtain the minimum adult frog population density estimate (or MAFD metric).
The MAFD metric was calculated as follows (Eq. 2):
$$MAFD=frac{Dc}{Maast Mpast Ms}$$
(2)
where MAFD – the minimum density of adult frogs on plot; Dc – the density of the calling males; Ma – the proportion of males that are vocalising during the peak activity (only from males present in waterbodies); Mp – the proportion of male total population present in waterbodies in the peak of the breeding season; Ms – the proportion of males in population (i.e., sex ratio). Taxon-specific parameters Ma, Mp, Ms were taken mainly from published studies (Table 8). Information on these parameters was generally scarce; thus, we did not find published Ma data on B.bombina and Rana. For these taxa we used Ma values from our unpublished visual observations of several ponds with excellent view of the water surface and a highly transparent water column. Regarding Ms, not all the adult females reproduce every year, and in terrestrial taxa (i.e. taxa not using waterbodies outside breeding season) the operational sex ratio at breeding waterbodies differs from the sex ratio in the whole adult population31,42. There are very limited data available for how much the operational sex ratio differs from the real sex ratio in surveyed taxa, so we ignored possible inter-species differences. We did reduce the published operational male proportions in terrestrial taxa – P.fuscus, both Rana, and H.arborea, by 15% (which was an average difference in31,43 for B.bufo and P.fuscus) to transform them into real proportions (i.e. Ms). For B.bufo we used published data on real sex ratio31. We did not find published data on B.bombina sex ratios and therefore we used information on other European Bombina species44,45,46.
We compared our MAFD estimates to visual observation records. In 58% of the plots at least one taxon had sufficient data from visual counts when they covered at least 75% of the surveyed waterbodies in the breeding season of a given taxon. We applied to observed frogs the following detection probabilities, which were inferred from other studies47,48,49,50 and personal observations: 0.75, when there were no obstacles to visual surface or underwater observations and active breeding behaviour (vocalising, moving, and mating) was observed; 0.50, when there were no obstacles to surface observations, but underwater visibility was poor or active behaviour absent; 0.20, when there were strong obstacles to visual observation; 0.10, for separate casual observations. The detection probability for egg masses was set as 0.9051. Egg counts were transferred into the respective numbers of males using the sex ratios from Table 8, and we also used sex ratios for visually counted adult Pelophylax spp. frogs with unknown sex (in contrast, typically only males are free moving in breeding aggregations of Bufo and Rana29, pers.obs.). We used Ma to transfer visual data into virtual calling males for each waterbody and then added undetected virtual males to waterbody calling male counts and used Eqs. 1 and 2 to obtain the new density estimate (NDE). The fraction of population covered by an MAFD estimate was calculated as the ratio of MAFD to NDE.
Source: Ecology - nature.com



