Avise, J. C. et al. Intraspecific phylogeography: the mitochondrial DNA bridge between population genetics and systematics. Annu. Rev. Ecol. Syst. 18, 489–522 (1987).
Avise, J. C. Phylogeography: the history and formation of species. (Harvard university press, 2000).
Zink, R. M., Pavlova, A., Drovetski, S. & Rohwer, S. Mitochondrial phylogeographies of five widespread Eurasian bird species. J. Ornitol. 149, 399–413, https://doi.org/10.1007/s10336-008-0276-z (2008).
Lavrov, D. V. & Pett, W. Animal mitochondrial DNA as we do not know it: mt-genome organization and evolution in nonbilaterian lineages. Genome Biol. Evol. 8, 2896–2913, https://doi.org/10.1093/gbe/evw195 (2016).
Aliabadian, M., Kaboli, M., Nijman, V. & Vences, M. Molecular identification of birds: performance of distance-based DNA barcoding in three genes to delimit parapatric species. PLoS One 4, e4119 (2009).
Hebert, P. D., Stoeckle, M. Y., Zemlak, T. S. & Francis, C. M. Identification of birds through DNA barcodes. PLoS Biol. 2, e312 (2004).
Johnsen, A. et al. DNA barcoding of Scandinavian birds reveals divergent lineages in trans-Atlantic species. J. Ornitol. 151, 565–578 (2010).
Kerr, K. C. et al. Comprehensive DNA barcode coverage of North American birds. Mol. Ecol. notes 7, 535–543 (2007).
Kerr, K. C. et al. Filling the gap-COI barcode resolution in eastern Palearctic birds. Front. Zool. 6, 29 (2009).
Stoeckle, M. Y. & Thaler, D. S. DNA barcoding works in practice but not in (neutral) theory. PLoS One 9, e100755 (2014).
Wang, E., Van Wijk, R. E., Braun, M. S. & Wink, M. Gene flow and genetic drift contribute to high genetic diversity with low phylogeographical structure in European hoopoes (Upupa epops). Mol. Phylogenet. Evol. 113, 113–125 (2017).
Carneiro de Melo Moura, C. et al. Pliocene origin,ice ages and postglacial population expansion have influenced a panmictic phylogeography of the European Bee-Eater Merops apiaster. J. Divers. 11, 12 (2019).
Pârâu, L. G., Frias-Soler, R. C. & Wink, M. High Genetic Diversity among Breeding Red-Backed Shrikes Lanius collurio in the Western Palearctic. Diversity 11, https://doi.org/10.3390/d11030031 (2019).
Rising, J. et al. Handbook of the Birds of the World Alive. (Lynx Edicions Barcelona, 2019).
Schmaljohann, H. et al. Proximate causes of avian protandry differ between subspecies with contrasting migration challenges. Behav. Ecol. 27, 321–331 (2015).
Bairlein, F., Eikenaar, C. & Schmaljohann, H. Routes to genes: unravelling the control of avian migration—an integrated approach using Northern Wheatear Oenanthe oenanthe as model organism. J. Ornitol. 156, 3–14, https://doi.org/10.1007/s10336-015-1224-3 (2015).
Bairlein, F. et al. Cross-hemisphere migration of a 25 g songbird. Biol. Lett., rsbl20111223 (2012).
Bairlein, F. Migratory birds under threat. Science 354, 547–548 (2016).
Kaboli, M., Aliabadian, M., Chamani, A., Pasquet, E. & Prodon, R. Morphological relationships of the Wheatears (genus Oenanthe). Russ. J. Ecol. 44, 251–259, https://doi.org/10.1134/s1067413613030168 (2013).
Svensson, L. Identification guide to European passerines. (L. Svensson, 1992).
Aliabadian, M., Kaboli, M., Prodon, R., Nijman, V. & Vences, M. Phylogeny of Palaearctic wheatears (genus Oenanthe)–congruence between morphometric and molecular data. Mol. Phylogenet. Evol. 42, 665–675, https://doi.org/10.1016/j.ympev.2006.08.018 (2007).
Kaboli, M., Aliabadian, M., Guillaumet, A., Roselaar, C. S. & Prodon, R. Ecomorphology of the wheatears (genus Oenanthe). Ibis 149, 792–805 (2007).
Förschler, M. I., Khoury, F., Bairlein, F. & Aliabadian, M. Phylogeny of the mourning wheatear Oenanthe lugens complex. Mol. Phylogenet. Evol. 56, 758–767, https://doi.org/10.1016/j.ympev.2010.03.022 (2010).
Outlaw, R. K., Voelker, G. & Bowie, R. C. Shall we chat? Evolutionary relationships in the genus Cercomela (Muscicapidae) and its relation to Oenanthe reveals extensive polyphyly among chats distributed in Africa, India and the Palearctic. Mol. Phylogenet. Evol. 55, 284–292, https://doi.org/10.1016/j.ympev.2009.09.023 (2010).
Alaei Kakhki, N. et al. Phylogeography of the Oenanthe hispanica-pleschanka-cypriaca complex (Aves, Muscicapidae: Saxicolinae): Diversification history of open-habitat specialists based on climate niche models, genetic data, and morphometric data. J. Zool. Sys. Evol. Res. 56, 408–427, https://doi.org/10.1111/jzs.12206 (2018).
Schweizer, M. & Shirihai, H. Phylogeny of the Oenanthe lugens complex (Aves, Muscicapidae: Saxicolinae): paraphyly of a morphologically cohesive group within a recent radiation of open-habitat chats. Mol. Phylogenet. Evol. 69, 450–461, https://doi.org/10.1016/j.ympev.2013.08.010 (2013).
Panov, E. N. Comparative ethology and molecular genetics as tools for phylogenetic reconstructions: The example of the genus. Oenanthe. Biol. Bull. 38, 809–820, https://doi.org/10.1134/s106235901108005x (2011).
Schweizer, M. et al. Parallel plumage colour evolution and introgressive hybridization in wheatears. J. Evol. Biol. 32, 100–110 (2019).
Arizaga, J., Schmaljohann, H. & Bairlein, F. Stopover behaviour and dominance: a case study of the Northern Wheatear Oenanthe oenanthe. Ardea 99, 157–165 (2011).
Corman, A.-M., Bairlein, F. & Schmaljohann, H. The nature of the migration route shapes physiological traits and aerodynamic properties in a migratory songbird. Behav. Ecol. Sociobiol. 68, 391–402 (2014).
Hobson, K. A. & Wassenaar, L. I. Tracking animal migration with stable isotopes. (Academic Press, 2018).
Desjardins, P. & Morais, R. Sequence and gene organization of the chicken mitochondrial genome: a novel gene order in higher vertebrates. J. Mol. Biol. 212, 599–634 (1990).
Jühling, F. et al. tRNAdb 2009: compilation of tRNA sequences and tRNA genes. Nucleic Acids Res. 37, D159–D162 (2008).
Watanabe, Y., Suematsu, T. & Ohtsuki, T. Losing the stem-loop structure from metazoan mitochondrial tRNAs and co-evolution of interacting factors. Front. Genet. 5, 109, https://doi.org/10.3389/fgene.2014.00109 (2014).
Gibb, G. C., Kardailsky, O., Kimball, R. T., Braun, E. L. & Penny, D. Mitochondrial genomes and avian phylogeny: complex characters and resolvability without explosive radiations. Mol. Biol. Evol. 24, 269–280, https://doi.org/10.1093/molbev/msl158 (2007).
Allcock, A. L., Cooke, I. R. & Strugnell, J. M. What can the mitochondrial genome reveal about higher-level phylogeny of the molluscan class Cephalopoda? Zool. J. Linn. Soc. 161, 573–586 (2011).
Cabrera, A. A. et al. Fin whale (Balaenoptera physalus) mitogenomics: A cautionary tale of defining sub-species from mitochondrial sequence monophyly. Mol. Phylogenet. Evol. 135, 86–97, https://doi.org/10.1016/j.ympev.2019.02.003 (2019).
Kumar, S. Patterns of nucleotide substitution in mitochondrial protein coding genes of vertebrates. Genetics 143, 537–548 (1996).
Braun, E. L. & Kimball, R. T. Examining basal avian divergences with mitochondrial sequences: model complexity, taxon sampling, and sequence length. Syst. Biol. 51, 614–625 (2002).
Duchêne, S., Archer, F. I., Vilstrup, J., Caballero, S. & Morin, P. A. Mitogenome phylogenetics: the impact of using single regions and partitioning schemes on topology, substitution rate and divergence time estimation. PloS one 6, e27138 (2011).
Leavitt, J. R., Hiatt, K. D., Whiting, M. F. & Song, H. Searching for the optimal data partitioning strategy in mitochondrial phylogenomics: a phylogeny of Acridoidea (Insecta: Orthoptera: Caelifera) as a case study. Mol. Phylogenet. Evol. 67, 494–508, https://doi.org/10.1016/j.ympev.2013.02.019 (2013).
Wang, N., Hosner, P. A., Liang, B., Braun, E. L. & Kimball, R. T. Historical relationships of three enigmatic phasianid genera (Aves: Galliformes) inferred using phylogenomic and mitogenomic data. Mol. Phylogenet. Evol. 109, 217–225, https://doi.org/10.1016/j.ympev.2017.01.006 (2017).
Richards, E. J., Brown, J. M., Barley, A. J., Chong, R. A. & Thomson, R. C. Variation across mitochondrial gene trees provides evidence for systematic error: How much gene tree variation is biological? Syst. Biol. 67, 847–860 (2018).
Kainer, D. & Lanfear, R. The effects of partitioning on phylogenetic inference. Mol. Biol. Evol. 32, 1611–1627 (2015).
Wink, M. Use of DNA markers to study bird migration. J. Ornitol. 147, 234–244, https://doi.org/10.1007/s10336-006-0065-5 (2006).
Cummings, M. P. & Meyer, A. Magic bullets and golden rules: data sampling in molecular phylogenetics. Zoology 108, 329–336 (2005).
Hillis, D. M. Inferring complex phytogenies. Nature 383, 130 (1996).
Sloan, D. B., Havird, J. C. & Sharbrough, J. The on-again, off-again relationship between mitochondrial genomes and species boundaries. Mol. Ecol. 26, 2212–2236, https://doi.org/10.1111/mec.13959 (2017).
Wink, M. Bird migration “Out of Africa”: The evolution of bird migration. Der Falke – Journal für Vogelbeobachter 60, 26–30 (2014).
Hewitt, G. M. Some genetic consequences of ice ages, and their role in divergence and speciation. Biol. J. Linn. Soc. 58, 247–276 (1996).
Hewitt, G. The genetic legacy of the Quaternary ice ages. Nature 405, 907–913, https://doi.org/10.1038/35016000 (2000).
Finlayson, C. et al. Ecological transitions — But for whom? A perspective from the Pleistocene. Palaeogeogr. Palaeoclimatol. Palaeoecol. 329-330, 1–9, https://doi.org/10.1016/j.palaeo.2011.04.002 (2012).
Frenzel, B. Atlas of paleoclimates and paleoenvironments of the Northern Hemisphere. (Geographical Research Institute, Hungarian Academy of Sciences, Budapest, Gustav Fischer Verlag, Stuttgart Jena New York., 1992).
Lundqvist, J. & Saarnisto, M. Summary of project IGCP-253. Quat. Int. 28, 9–18 (1995).
Taberlet, P., Fumagalli, L., Wust-Saucy, A. G. & Cosson, J. F. Comparative phylogeography and postglacial colonization routes in Europe. Mol. Ecol. 7, 453–464 (1998).
Irwin, D. E., Rubtsov, A. S. & Panov, E. N. Mitochondrial introgression and replacement between yellowhammers (Emberiza citrinella) and pine buntings (Emberiza leucocephalos)(Aves: Passeriformes). Biol. J. Linn. Soc. 98, 422–438 (2009).
Semenov, G. A., Koblik, E. A., Red’kin, Y. A. & Badyaev, A. V. Extensive phenotypic diversification coexists with little genetic divergence and a lack of population structure in the White Wagtail subspecies complex (Motacilla alba). J. Evol. Biol. 31, 1093–1108, https://doi.org/10.1111/jeb.13305 (2018).
Johnsen, A., Kearns, A. M., Omland, K. E. & Anmarkrud, J. A. Sequencing of the complete mitochondrial genome of the common raven Corvus corax (Aves: Corvidae) confirms mitogenome-wide deep lineages and a paraphyletic relationship with the Chihuahuan raven C. cryptoleucus. PLoS One 12, e0187316, https://doi.org/10.1371/journal.pone.0187316 (2017).
Kearns, A. M. et al. Genomic evidence of speciation reversal in ravens. Nat. Commun. 9, https://doi.org/10.1038/s41467-018-03294-w (2018).
Palacios, C. et al. Shallow genetic divergence and distinct phenotypic differences between two Andean hummingbirds: Speciation with gene flow? The Auk 136, ukz046 (2019).
Wang, K. et al. Incomplete lineage sorting rather than hybridization explains the inconsistent phylogeny of the wisent. Commun. Biol. 1, 169, https://doi.org/10.1038/s42003-018-0176-6 (2018).
Wang, W. et al. Past hybridization between two East Asian long-tailed tits (Aegithalos bonvaloti and A. fuliginosus). Front. Zool. 11, 40 (2014).
McKay, B. D. & Zink, R. M. The causes of mitochondrial DNA gene tree paraphyly in birds. Mol. Phylogenet. Evol. 54, 647–650 (2010).
Choleva, L. et al. Distinguishing between incomplete lineage sorting and genomic introgressions: complete fixation of allospecific mitochondrial DNA in a sexually reproducing fish (Cobitis; Teleostei), despite clonal reproduction of hybrids. PLoS One 9, e80641, https://doi.org/10.1371/journal.pone.0080641 (2014).
He, S. et al. An examination of introgression and incomplete lineage sorting among three closely related species of chocolate-dipped damselfish (genus: Chromis). Ecol. Evol. 9, 5468–5478, https://doi.org/10.1002/ece3.5142 (2019).
Li, S., Luo, A., Li, G. & Li, W. Complete mitochondrial genome of the isabelline wheatear Oenanthe isabellina (Passeriformes, Muscicapidae). Mitochondrial DNA Part B 1, 355–356, https://doi.org/10.1080/23802359.2016.1167641 (2016).
Sambrook, J., Fritsch, E. & Maniatis, T. Molecular cloning: A laboratory manual+ Cold Spring Harbor. (New York: Cold spring harbor laboratory press, 1989).
van Oosten, H. H., Mueller, J. C., Ottenburghs, J., Both, C. & Kempenaers, B. Genetic structure among remnant populations of a migratory passerine, the Northern Wheatear Oenanthe oenanthe. Ibis 158, 857–867 (2016).
Randler, C. et al. Phylogeography, pre-zygotic isolation and taxonomic status in the endemic Cyprus Wheatear Oenanthe cypriaca. J. Ornitol. 153, 303–312 (2012).
Hahn, C., Bachmann, L. & Chevreux, B. Reconstructing mitochondrial genomes directly from genomic next-generation sequencing reads—a baiting and iterative mapping approach. Nucleic. Acids. Res. 41, e129–e129 (2013).
Chevreux, B., Wetter, T. & Suhai, S. in German conference on bioinformatics. 45-56 (Citeseer).
Lowe, T. M. & Chan, P. P. tRNAscan-SE On-line: integrating search and context for analysis of transfer RNA genes. Nucleic Acids Res. 44, W54–57, https://doi.org/10.1093/nar/gkw413 (2016).
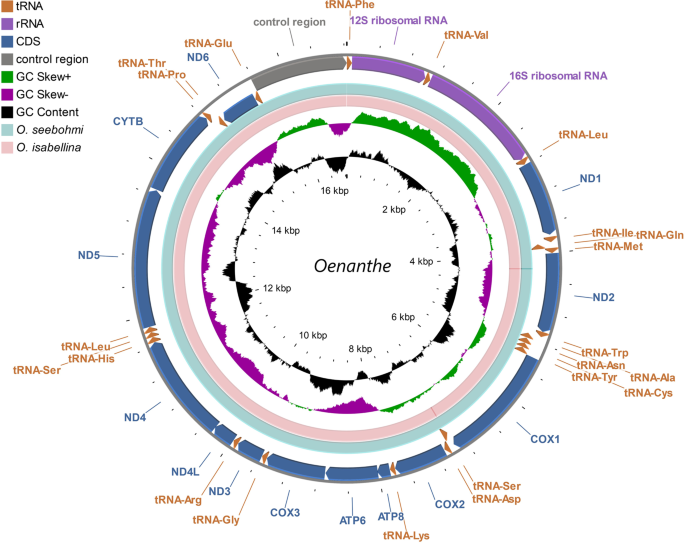
Stothard, P. & Wishart, D. S. Circular genome visualization and exploration using CGView. Bioinformatics 21, 537–539 (2004).
Katoh, K. & Standley, D. M. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evol. 30, 772–780 (2013).
Vaidya, G., Lohman, D. J. & Meier, R. J. C. SequenceMatrix: concatenation software for the fast assembly of multi‐gene datasets with character set and codon information. Cladistics 27, 171–180 (2011).
Lanfear, R., Frandsen, P. B., Wright, A. M., Senfeld, T. & Calcott, B. PartitionFinder 2: new methods for selecting partitioned models of evolution for molecular and morphological phylogenetic analyses. Mol. Biol. Evol. 34, 772–773 (2016).
Lanfear, R., Calcott, B., Ho, S. Y. & Guindon, S. PartitionFinder: combined selection of partitioning schemes and substitution models for phylogenetic analyses. Mol. Biol. Evol. 29, 1695–1701 (2012).
Ronquist, F. et al. MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 61, 539–542 (2012).
Altekar, G., Dwarkadas, S., Huelsenbeck, J. P. & Ronquist, F. Parallel metropolis coupled Markov chain Monte Carlo for Bayesian phylogenetic inference. Bioinformatics 20, 407–415 (2004).
Drummond, A. J. & Rambaut, A. BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol. Biol. 7, 214 (2007).
Suchard, M. A. & Rambaut, A. Many-core algorithms for statistical phylogenetics. Bioinformatics 25, 1370–1376 (2009).
Lanave, C., Preparata, G., Sacone, C. & Serio, G. A new method for calculating evolutionary substitution rates. J. Mol. Evol. 20, 86–93 (1984).
Weir, J. T. & Schluter, D. Calibrating the avian molecular clock. Mol. Ecol. 17, 2321–2328, https://doi.org/10.1111/j.1365-294X.2008.03742.x (2008).
Excoffier, L. & Lischer, H. E. Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol. Biol. Resour. 10, 564–567 (2010).
Librado, P. & Rozas, J. DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25, 1451–1452 (2009).
Source: Ecology - nature.com


