In the present study, mitochondrial and nuclear diversity was examined in feral raccoons introduced into Hokkaido, Japan. This is the first report to study raccoon population dynamics based on genetic differentiation in Japan and to compare genetic characteristics among this animal in Japan with an original habitat in North America.
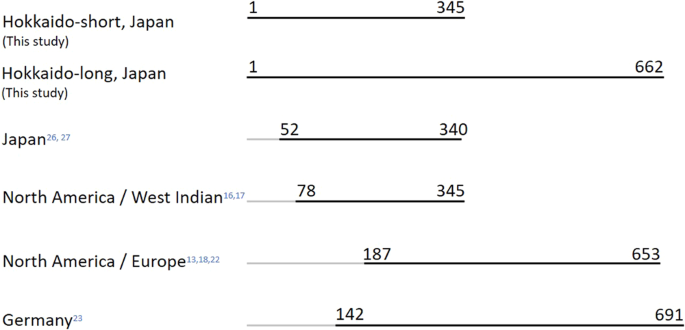
The 662-bp sequences detected in this study covered fragments identified in previous studies, including 289 bp in Japan27, 268 bp in North America and West Indian islands16,17, and also 467 bp in eastern North America18 (Fig. 1). The sequences also partially covered 521 bp out of a 550 bp fragment from Germany23. Here, seven haplotypes were detected from 526 samples. Five of these were reported previously in central Hokkaido27. When adding RMT06, which had been found only in the study by Takada-Matsuzaki et al.27 but not in the present study, founder events in Hokkaido were established from at least eight female haplotypes (Fig. 2A). This could be an underestimation, given that founder phylogenetic diversity can diminish during changing demographic periods based on the number of founder females and survivorship. Compared with reports in native habitats in Northeastern America, where 76 haplotypes were detected from 311 samples18, the number of haplotypes in Hokkaido is small. Such low mitochondrial diversity was also reported in other introduced areas in Europe, with two haplotypes identified from 58 samples in Spain13, six haplotypes from 193 samples in Germany23, and four haplotypes from 72 samples from the border area in Poland, Czech Republic, and Germany22. Rapid expansion from small populations can involve serial bottlenecking with progressive loss of allelic diversity, so that populations in more recently colonized locations could contain less genetic diversity31.
Four haplotypes out of eight identified in this study were also detected in North America18 and five haplotypes out of the eight were also detected in the other locations within Japan32, indicating that raccoons from the same origin group in North America were introduced at several remote places in Japan as founder events. Another possibility is that re-introduction (as a second release in the prefectures) from the initial introduced regions into the non-habitat area, happened after the first founder event as discussed by Takada-Matsuzaki et al.27 Haplotype NA matched haplotype PLO2, which was mentioned in Cullingham’s study18. PLO2 is the most common raccoon haplotype, is widespread in North America, and has also been reported in non-native areas, such as Germany, Spain, Poland, and Czech Republic13,22,23. Haplotype NA was detected in introduced areas across countries, which strongly suggests that the same genetic population of this haplotype was managed under captivity from wild populations in North America, exported as pets or as a fur-bearing animal and was introduced into non-native regions.
In this study, 326 individuals were divided into six groups based on mtDNA polymorphism, STRUCTURE analysis, and habitat regions, by use of the municipality as a unit. We note that this classification might not be biologically relevant for wild animals that move regardless of the frontiers between municipalities. In Hokkaido, however, most eradication programs of raccoons have been implemented by each municipality. Therefore, this study focused more on classifying management units, rather than on finding the ecological boundary between sub-populations.
Genetic diversity at these ten microsatellite loci in Hokkaido raccoons is low in Na though it was at the same level in Ho and He when compared to that in a native habitat of North America (in Chicago28,33 and Illinois34) (Table 2). When genetic variability defined by clusters was evaluated in the six management units (Table 3), Ar, Ho, and He were high compared to Germany where feral raccoons are widespread throughout the country24. Values were low in six clusters of 407 feral German raccoons: Ar: 3.6–4.9, Ho: 0.52–0.64, and He: 0.53–0.65. These differences demonstrate that nuclear genetic diversity in Hokkaido has stayed polymorphic at the same level as native habitat, in contrast to other invaded habitats.
In other raccoon-invaded areas, genetic diversity at the nuclear level in Spain was lower than in Missouri, a native area13. Raccoons were first reported in Spain relatively recently, in 2003. The microsatellite analysis consisted of 58 samples and showed only two mtDNA haplotypes captured in the limited area of Madrid. Therefore, diversity may be lower than observed in our study and in reports from the United States. In contrast, raccoons have been in Germany much longer than in Spain. There, raccoons were introduced in the 1930s, but the populations were derived from two separate founding events and one additional escape event24. These small numbers of founding events likely led to low polymorphisms seen in Germany24. The high genetic polymorphisms observed in this study indicate that the founder event in Hokkaido was relatively complex compared to that in Spain or Germany. The mtDNA of raccoons in Hokkaido showed allopatric, whereas the genome DNA showed very low pairwise FST values between any two management units regardless of the distance of the two units. Almost all the FST values (0.005–0.067) in the current study was lower than the lowest value observed in Germany (0.049)23. This indicates that several maternal populations, with different genetic backgrounds, were introduced into different regions in Hokkaido. However, these original populations were closely related to each other, resulting in limited differentiation among Units even 40 years after raccoons were introduced.
In Japan, raccoons were mainly imported as pets and introduced into the wild afterwards35. To our best knowledge, there are no documents recording when and how many raccoons were imported into Japan7, though the results of this study reflect that founder populations in Hokkaido likely derived from genetically related individuals from a captive breeder in North America or the main island of Japan. When allelic richness was compared among the six management units in Hokkaido, Unit2 and Unit3 showed relatively high genetic diversity and also highly diverse mtDNA (Table 3). Unit3, covering Sorachi, Ishikari, and Iburi subprefectures (the most human populated regions), which includes Sapporo, the largest city in Hokkaido. Unit2 has the second largest city, Asahikawa, suggesting that raccoons from multiple haplotypes were released into highly populated regions. Historical evidence demonstrates that Ishikari, Sorachi, and Kamikawa subprefectures were regions where feral raccoons or their traces were detected in the 1990s, earlier than in other regions (Hokkaido governmental reports and Ikeda7). A report showing that national parks with high surrounding human population density had significantly more alien species36 also supports our findings. Additionally, in Hokkaido, genetic polymorphism was varied but conserved, not only in highly populated areas, but also in regions throughout the island. This result suggests that several founder females with different haplotypes were introduced sympatrically and/or allopatrically, and cross-municipality migrating has resulted in the genetic diversity observed in Hokkaido.
Ikeda6 reported that the naturalization of raccoons in Hokkaido was due to multiple escapes and abandonment of pets. They also reported that additional human-induced transport and secondary releases have allowed habitat expansion in more remote locations within Hokkaido. Raccoons tend to disperse along the rivers and water passages37. Females are often philopatric, while males are more likely to disperse37,38. In the native habitat, the majority of raccoons relocate less than 5 km39, meanwhile they sometimes disperse more than 20 km in the absence of major landscape barriers30. Genetic analysis here suggests that geographical barriers prevent migration of feral individuals and reduce genetic admixture between some populations. The mtDNA haplotype of Unit1 and a part of Unit4 were the same; genetic structure in microsatellite clusters was different between these two units, indicating that there was less genetic intercrossing between north and southwest regions in Hokkaido. Geographical distances between these regions also support that there has been less genetic admixture between these units after individuals were introduced from the same origin. Unit2 was limited to Kamikawa and Sorachi Subprefectures, and Unit6 in Tokachi Subprefecture, which are neighboring regions. Although mtDNA haplotypes were different between these two units, STRUCTURE analysis inferred possible genetic admixture, suggesting that male individuals might have been dispersed among these regions.
Unit5 in Hidaka Subprefecture and Unit6 are also neighboring regions and have the same mtDNA haplotype constitution. However, STRUCTURE results indicated that there was less individual contact. There is approximately 150 km of the Hidaka mountain range between Hidaka and Tokachi, which could have prevented genetic mixture by serving as a geographical barrier, resulting in each population having maintained their own genetic structure. It is thus important for neighboring areas to cooperate with each other to control the spread of invasive raccoons. In the case of Hidaka and Tokachi, there could be effective management considering the geographical barriers19. Although human-induced re-introduction and transport were reported at the beginning of raccoon naturalization, the Invasive Alien Species Act of 2005 strictly regulates transportation of living animals. In this situation, biological information, such as geographical isolation or populational genetics, will be helpful for effective eradication and control19.
Source: Ecology - nature.com


