Study system
This study was carried out in the region of Montpellier, southern France, with insects from natural populations collected in fig trees present at the CEFE (“Centre d’Ecologie Fonctionnelle et Evolutive”) experimental garden (43°38′19″N, 3°51′49″E) in Montpellier, France, and from natural populations less than 40 km distant from Montpellier.
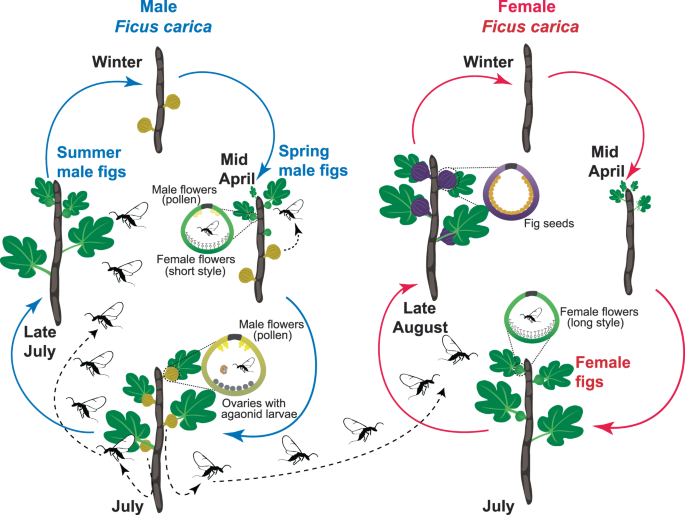
Our model system is the mutualistic interaction between the agaonid Blastophaga psenes L. (Hymenoptera, Chalcidoidea, Agaonidae) and its exclusive host, the Mediterranean Ficus species, i.e. Ficus carica (subgenus Ficus, section Ficus, subsection Ficus). This pollinating fig wasp species has two generations per year coinciding with the flowering of male trees first in April (“spring male figs”) and then in July (“summer male figs”) (Fig. 1). In contrast, female trees flower only once a year, in July, and thus partially synchronously with summer male figs (Fig. 1). Detailed life cycles of both Blastophaga psenes and Ficus carica are presented in Fig. 1 and in50,53.
Preparation of odour sources
As for all the analysis and behavioural tests, odour preparations were conducted at the “Platform for Chemical Analyses in Ecology” (PACE, Montpellier) technical facilities of the LabEx CeMEB (“Centre Méditerranéen pour l’Environnement et la Biodiversité”, Montpellier, France). For the GC-EAD recordings, VOCs of receptive summer male figs and female figs were collected using classical adsorption-desorption headspace technique30,54,55. Three groups of 20 to 30 receptive figs were collected haphazardly from both male and female trees and directly enclosed in polyethylene terephthalate bags. For each bag, traps containing 30 mg of Alltech Super Q adsorbent (ARS Inc., Gainesville, FL, USA) were placed at the end of tube from which air was drawn in. Airflow was maintained through the bags by two pumps (KNF, Neuberger, Freiburg, Germany). Air pushed into the bag by a polytetrafluoroethylene (PTFE) tube was filtered using activated charcoal. The entrance and exit flow rates were regulated by flowmeters at 300 and 200 ml·min−1, respectively, to create a positive pressure inside the bag and thereby prevent contamination from the environment. The collection duration was 4 h. Each trap was eluted with 150 μl of hexane (Sigma Aldrich, Munich, Germany, purity >99%) and the three extracted samples were pooled together and stored at −20 °C. In addition to these odour samples, synthetic compounds were used during the electrophysiological recordings to validate the antennal responses.
For the behavioural tests, summer male figs, female figs, and synthetic compounds, singly or in blends, were used. Fresh male and female figs were collected in the field and used within 2 h after collection for the behavioural tests. For these bioassays, a single fig was used for each test. Furthermore, four synthetic compounds were chosen based on their activity on insect antennae (see below) and their constant occurrence in the volatile profile of receptive figs of F. carica29,30. With these compounds, seven blends were prepared with different compound proportions (Table 1). The composition of the blend 1 (B1) was based on the average proportion and quantities of the four synthetic compounds emitted by the three types of figs (Table 1), with the constraint that some VOCs were not available commercially as pure compound. In order to investigate the importance for pollinator attraction of each compound in the blend, in the following six other blends we reduced or increased the proportions of the different VOCs. To deliver the odours in the experimental set-up (Supplementary, Fig. S1), pure synthetic compounds were added into a glass insert of 400 μl, which was placed into a 1.5 ml vial sealed with a polytetrafluoroethylene/rubber septum (Chromoptic, Courtaboeuf, France). A micro-capillary tube made of fused silica (Agilent technologies, Redmond, USA), 40 mm long and 0.53 mm internal diameter (ID), was inserted through the septum. The diameter and length of the capillary tube were calibrated to release a controlled amount of VOCs corresponding to the mean release rates of one receptive fig. For that, vials were positioned inside glass containers of 500 ml and a diameter of 100 mm that were connected to a continuous airflow of 200 ml.min−1 and maintained inside an oven at 25 °C. Vials were weighed regularly during 2 months using a microbalance (MC5, Sartorius, Goettingen, Germany) to determine the diffusion rate (ng.min−1). Preliminary tests revealed that diffusion of the VOCs using these dispensers is only stable after 10 days (Proffit et al., unpublished data).
Electrophysiology on B. psenes
GC-EAD recordings were conducted on a gas chromatograph-flame ionization detector (GC-FID, CP-3800, Varian, Palo Alto, USA) equipped with an optima 5-MS capillary column (30 m, 0.25 mm ID, 0.25 μm film thickness, Machery-Nagel, Düren, Germany) coupled to an electroantennography detector setup (EAD, Syntech IDAC-2, Kirchzarten, Germany). Four μl of either receptive fig odour or synthetic mix solution were injected into the GC-FID. The injector was heated to 250 °C, with a 1:4 split ratio to inject the compounds into the column. Oven temperature was held at 50 °C for 1 minute, increased from 50 °C to 100 °C at a rate of 9 °C.min−1 then from 100 to 140 °C at a rate of 8.1 °C.min−1, then from 140 °C to 190 °C at a rate of 7.2 °C min−1, then from 190 °C to 210 °C at a rate of 20 °C.min−1 and finally the temperature was held at 210 °C during 50 seconds. The carrier gas used was helium at 1 ml·min−1. The effluent was split equally into two deactivated fused silica capillary columns (100 cm × 0.25 mm), one leading to the FID (270 °C) and one into a heated EAD port (200 °C) (transfer line, Syntech, Kirchzarten, Germany). For the EAD, wasp heads were cut at their base. Head base and the tip of one antenna were mounted between two glass capillary tubes filled with insect Ringer solution (6.0 g·l−1 NaCl, 0.4 g·l−1, KCl, 0.27 g·l−1, CaCl2 and 3.20 g·l−1 of sodium lactate) and connected to silver wires. Electrophysiological measurements were conducted separately with the antennae of seven adult female wasps for each odour source tested. A compound was considered to be EAD-active when it elicited an unequivocal depolarization response in four antennae out of seven. In addition, the activity of VOCs on pollinator antennae was confirmed using synthetic standards.
Volatile collections and chemical analysis
Headspace collections of VOCs of spring male (N = 6), summer male and female receptive figs (N = 4 for each), and of all the dispensers used for the behavioural test were conducted. For these collections, either receptive figs or an odour dispenser were placed into a glass container of 500 ml for 30 minutes before collection. Air pushed into the glass container was filtered using activated charcoal at a flow rate of 200 ml·min1. The same amount of air was drawn out of the container through an adsorbent trap compatible with a thermal desorption system, consisting of an external glass tube (length: 60 mm and 6 mm O.D., Gerstell, Mulheim, Germany) filled with 80 mg of Tenax-TA and 40 mg of Carbotrap (60–80 and 20–40 mesh respectively, Sigma-Aldrich, Munich, Germany). Odour collections lasted 10 min for the synthetic compounds and 30 min for the figs. All adsorbent traps were sealed with lids on both side openings and stored at −20 °C until further use.
Chemical analyses were conducted using a method similar to that of Souto-Vilarós et al.18. We used a gas chromatograph (GC, Trace 1310, Thermo Scientific, Milan, Italy) coupled to a mass spectrometer (ISQ QD Single Quadrupole, Thermo Scientific, Milan, Italy). The column used was an Optima 5-MS capillary column (30 m, 0.25 mm ID, 0.25 μm film thickness, Machery-Nagel, Düren, Germany). Absorbent traps were handled with a Multi Purpose Sampler (Gerstell, Mülheim, Germany) and desorbed with a double stage desorption system, composed of a Thermal Desorption Unit (TDU) and a Cold Injection System (CIS) (Gerstell, Mülheim, Germany). First, the injector was splitless with a temperature of 250 °C on the CIS trap cooled at −80 °C by liquid nitrogen. Then, the CIS trap was heated to 250 °C with a 1:4 split ratio to inject the compounds in the column. We used helium at 1 ml·min−1 as a carrier gas. Oven temperature was held at 40 °C for 3 minutes, increased from 40 °C to 220 °C at a rate of 5 °C·min−1 and from 220 to 250 °C at 10 °C·min−1, and finally held for 2 minutes at 250 °C. The temperature of the transfer line and the ion source of the mass spectrometer was 250 °C and 200 °C respectively. The acquisition was at 70 eV ionisation energy, from 38 m/z to 350 m/z. We used Xcalibur TM 266 software (Thermo Scientifc TM, Milan, Italy) for data processing. Retention times of a series of n-alkanes (Alkanes standard solution, 04070, Sigma Aldrich, Munich, Germany) were used to convert retention times into a retention index. Compound identification was based on computer matching of mass spectra and retention indices with reference compounds. In addition, to quantify precisely the emission rate of the VOCs detected by the pollinator in each sample, known amounts of these different reference compounds (100, 50, 20 and 2 ng) were injected into an adsorbent trap and analysed in the GC-MS system using the same method. Their mean peak areas were used for calibration. In addition, the stereochemistry of linalool was determined using the same analytic method as indicated above in the same GC-MS equipped with a β-cyclodextrin chiral capillary column (Cyclosil-B, 30 m–0.25 mm i.d., 0.25 μm film thickness, Agilent J&W columns, USA).
Behavioural experiments
We tested whether females of B. psenes were attracted to VOCs emitted by receptive figs of F. carica and by several synthetic compounds, using a glass Y-tube olfactometer in which only chemical cues were presented to wasps (Supplementary, Fig. S1)6,18,23. Bioassays were conducted from 2016 to 2019 under laboratory conditions at the temperature of 25.75 ± 0.10 °C and relative humidity of 51.52 ± 1.60%. Y-tubes were in glass (40 mm in diameter), each lateral arm was 200 mm long and the central arm was 150 mm long. Odour sources were placed in glass containers of 500 ml and connected to each lateral arm of the Y-tube. Air was purified with activated charcoal, humidified with distilled water and blown into the glass containers (200 ml·min−1 per arm). Y-tubes and glass containers were changed and cleaned with acetone after each trial in order to remove any chemical traces left by the insects. To avoid a directional bias, the positions of odour sources were inverted between the two arms in each successive trial. Treatments were alternated to be able to compare wasp choice frequencies between treatments. For the bioassays, between 42 and 60 fig wasps were tested per treatment. Air was blown for 1 min prior to insect introduction into the olfactometer. During ten minutes, the choice made by each individual was recorded. We considered that wasps did not choose when they stayed motionless in the departure section and/or the central arm before the bifurcation of the olfactometer after these ten min. These wasps were not taken into account in the statistical analyses.
Newly emerging adult female wasps were collected from mature figs taken haphazardly from different individual male trees. Because of their very short lifespan outside the fig (less than 24 h), individuals of B. psenes were tested shortly after their exit. Each day a maximum of 25 individuals were tested per treatment. All tested wasps were naïve to the odour sources presented in our bioassays.
Data analysis
All the data analyses were performed in R (v. 3.5.3; R Development Core Team; http://www.R-project.org) using multivariate analysis incorporated in the Vegan package56. As variation in the relative proportions of all the VOCs emitted by receptive figs of F. carica was compared in a previous study29, we only focused on comparison among the three types of receptive figs of relative proportions of the four VOCs used by the pollinator. Data were standardized before the analyses and a data matrix of pairwise Bray-Curtis dissimilarity indices between samples was built. Non-metric multi-dimensional scaling (NMDS) was used to visualize similarities among the samples by finding the best two-dimensional representation of the distance matrix. A Permutational Multivariate Analysis of Variance (PERMANOVA) based on 999 permutations was used to test the null hypothesis of no centroid (i.e. mean) difference in on the relative proportions of the four VOCs among the three different types of figs, as well as in pairwise comparisons. The difference of dispersion (i.e. variance) in the relative proportions of these VOCs among the three different types of figs, and also in pairwise comparisons, was tested using a multivariate analogue of Levene′s test for homogeneity of variance. Finally, we performed an ANOVA to compare the total amounts of VOCs emitted by receptive figs of the three types. For pairwise comparisons, P-values were adjusted for multiple comparisons using the FDR method57,52.
For all dual-choice bioassays in the Y-tube olfactometer, two-tailed exact binomial tests were used to test the null hypothesis that the same number of wasps was attracted to both odour sources.
Source: Ecology - nature.com


