Violacein toxicity in larva and adult C. elegans
To gain a deeper understanding of the effects of violacein on C. elegans physiology and development, we made a violacein-producing E. coli strain OP50 by transforming it with pCOM10vio plasmid, which expresses the five genes of the violacein operon under the PalkB promoter, commonly used in E. coli for inducible expression17 (henceforth called OP50-vio). Expression of violacein was induced at 20 °C, with a purple hue visually apparent when OP50-vio is grown on standard nematode growth media (NGM) (Fig. 1A). Based on previous observations with DH5α (unpublished observation), OP50-vio is expected to produce comparable levels of violacein as bacteria found in the wild19. Wild-type N2 worms grown on normal OP50 proceed through several larval stages from L1 to L4 and grow in size before becoming an adult worm after 4 days, at which point they laid their own eggs. However, worms grown on OP50-vio showed little growth and eventually experienced a developmental arrest in early larval stages (Fig. 1B,C). Thus, violacein has a profoundly negative impact on C. elegans growth and development.
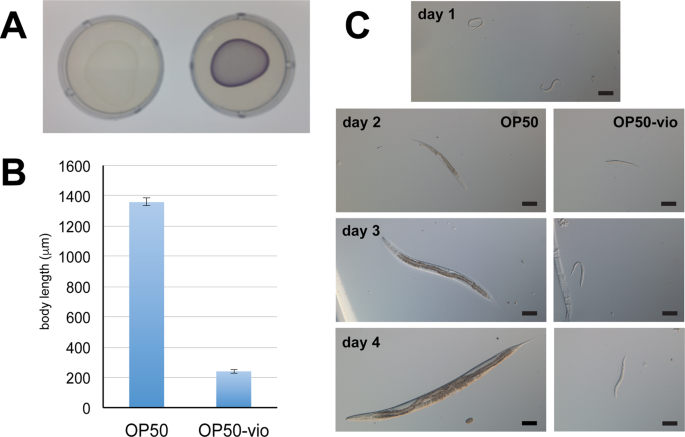
Violacein stunts the growth and development of C. elegans. (A) Lawn of E. coli strain OP50 (left) and violacein-expressing OP50 (OP50-vio, right). (B) Body length of worms grown on OP50 and OP50-vio from L1 larvae stage for 4 days. (C) Development of worms grown on OP50 and OP50-vio. Day 1 image show L1 synchronized worms that has never been fed. Scale bar = 100 μm.
Violacein also imparted serious consequences to the adult worm. When adults were exposed to violacein overnight, we observed its accumulation in the intestine, along with distension of the gut lumen and general intestinal morphology defects (Fig. 2A,B), which is similar with previous reports10. Worms showed a smaller and shriveled appearance in general.
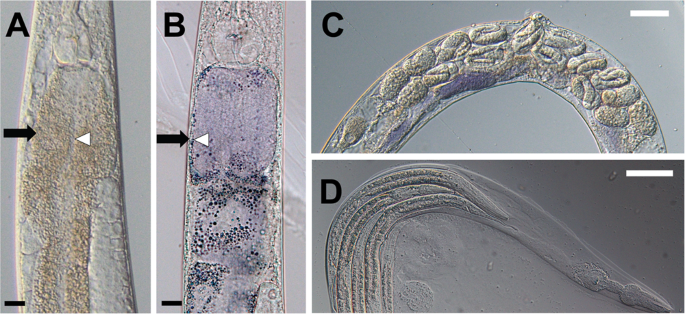
Violacein induces multiple defects in the adult hermaphrodite mothers. (A,B) Adult nematodes exposed to violacein develop intestinal defects. (A) shows OP50-fed worms and (B) shows worms fed OP50-vio. Intestinal walls (black arrows) are thinner and lumen width (white arrows) is wider in OP50-vio exposed adult animals. (C) Adult hermaphrodite mothers in OP50-vio display late-stage eggs within their body. (D) Internally hatched larvae worms inside a dead mother’s remaining cuticle in OP50-vio. Black scale bar indicate 20 μm, white scale bar 50 μm.
In addition to these intestinal effects, consumption of violacein inhibited the mother worm from laying her eggs, who instead held them within her body. Under normal developmental processes, C. elegans oocytes are fertilized internally, and the mother lays the eggs through her vulva when the embryo reaches between the 16 to 32-cell stage. However, in violacein-treated mothers late stage embryos can be seen inside the mother worms’ body (Fig. 2C), and eventually we observed internal hatching of the embryos in many of the mothers (Fig. 2D). In this phenotype, commonly seen when gravid C. elegans mothers are exposed to adverse environmental conditions15,20,21,22,23,24,25, some of the embryos that are not laid will hatch internally, and the larvae will begin to feed and grow within the mother’s body. This results in the death of the mother and a “bag of worms” phenotype in which larvae are surrounded by the mother’s remaining cuticle (Fig. 2D). Under such conditions, the larvae continue to grow and finally break open the mother’s cuticle before being released into the surrounding environment.
Previous reports demonstrated that violacein decreased the C. elegans lifespan significantly10,11. However, neither of those studies distinguished whether violacein toxicity or internal hatching was directly responsible for worm death. Consequently, we evaluated C. elegans’ survival on OP50-vio with or without addition of 5-fluoro-2′-deoxyuridine (FUDR), a chemical that prevents internal hatching by inhibiting cell division and subsequent development of the embryos. As shown in Fig. 3A, the presence of FUDR significantly increased survival of the worms, suggesting that internal hatching is a major cause of violacein-induced death (Fig. 3A). This is not to say that this is the only form of toxicity, though, as the survival rates with FUDR treatment were still much lower for worms grown on OP50-vio than normal OP50 (Fig. 3A). Thus, violacein-induced death is due to both internal hatching and general toxicity.
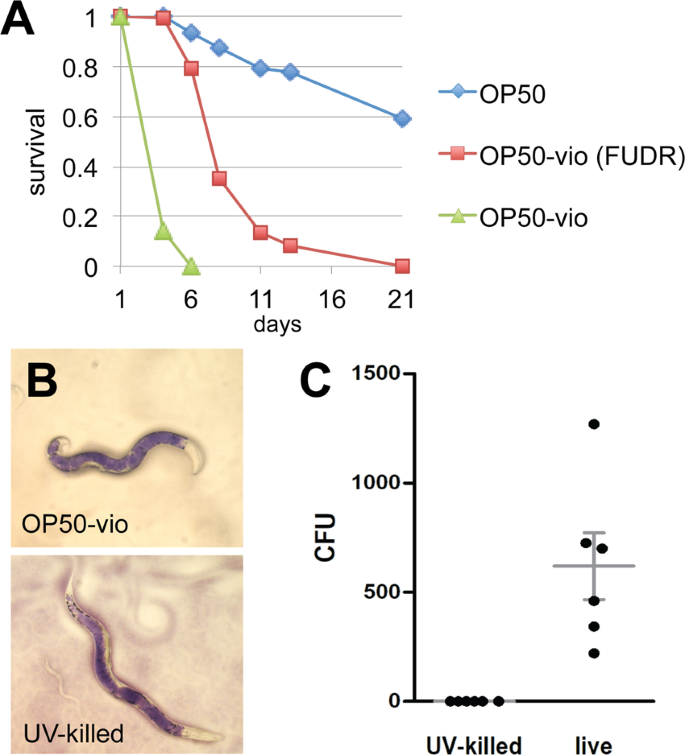
Violacein shortens lifespan independent of internal hatching and intestinal colonization. (A) Survival of C. elegans adults in OP50-vio treated with or without FUDR treatment. C. elegans worms normally die within 5–6 days on OP50-vio due to internal hatching, but worms treated with the sterilizing agent FUDR can survive up to 20 days. Representative graph from 3 trials; n ranges from 50 to 136. (B) Violacein accumulates in the intestine of both OP50-vio and UV-killed OP50-vio cultured worms, indicating defect in passing contents out of the intestine, rather than active colonization of bacteria. (C) UV-killed OP50-vio does not colonize the intestine. Colony counts of bacteria inside the intestinal lumen of each worm with either OP50-vio or UV-killed OP50-vio (n = 6).
Violacein-producing E. coli was previously shown to colonize and accumulate in the worm intestine, which would contribute to toxicity to the worm10. To evaluate whether colonization of OP50-vio is required for death, we UV-irradiated OP50-vio to kill the bacteria. When we fed adult worms the UV-killed OP50-vio, we still observed accumulation of violacein pigment in the intestine (Fig. 3B), even though we confirmed that the accumulated bacteria were dead (Fig. 3C). Moreover, the survival rate of C. elegans when provided UV-killed OP50-vio was indistinguishable from the live OP50-vio (Fig. S1). This demonstrates that at least when expressed in OP50, death is a result of violacein itself rather than the colonization of violacein-producing bacteria in the gut.
Internal hatching ameliorates the developmental effects of violacein
In our evaluation of violacein toxicity, an interesting observation was made. Although violacein completely arrests larval development so that none of the larvae can reach adulthood (Fig. 1), we noticed that in some of the old OP50-vio plates in which adult worms were placed more than a week before, the adults had already died; however, a small portion of adult worms from the next generation had grown up. This was confounding as we already showed that young larvae cannot develop and survive in violacein (Fig. 1B,C). Since we knew that C. elegans mothers in violacein allow their larvae to hatch internally (Figs. 2C,D), we hypothesized that internal hatching conferred some benefit to allow the larvae to survive and grow in the presence of violacein.
To test whether the presence of the mother worm could mitigate violacein’s effect on development, we placed either young adult mother worms or just their eggs directly in OP50-vio for 10 days and compared the growth of the resulting larvae. We found larvae from the laid eggs (N2 egg) did not grow as well as those that grew in the presence of their mothers (N2), but the difference was very subtle and statistically insignificant (Fig. 4). Because it takes several hours for the worms to stop laying eggs in response to violacein, most eggs are laid and only a small portion of young will hatch internally. To ensure that most of the larvae hatch internally within the mother’s body, we measured the growth of young from unc-54 mutant mothers in violacein. Mutants of the myosin heavy chain gene unc-54 lack functioning muscle, including the vulva muscle, meaning 100% of the embryos develop within the mother and hatch internally26. Although they are severely defective in movement, they grow quite normally to adulthood, albeit slightly shorter in length (Fig. S3). The larvae from unc-54 mutant mothers, on average, grew better in the presence of violacein than larvae from either the wild-type eggs or wild-type mothers (Fig. 4). Particularly, some of the unc-54 larvae grew substantially longer, i.e., to adulthood or near adulthood, which was rarely seen in N2 larvae and never observed in N2 eggs (Table 1). To verify that this effect is due to internal hatching, we prepared eggs from unc-54 worms to hatch outside the mother in OP50-vio. Eggs hatched outside the mother displayed stunted growth comparable to N2 eggs (Fig. 4). This indicates that internal hatching, which is induced in the presence of violacein, provides a significant benefit for some of the larvae against the bacterial toxin.
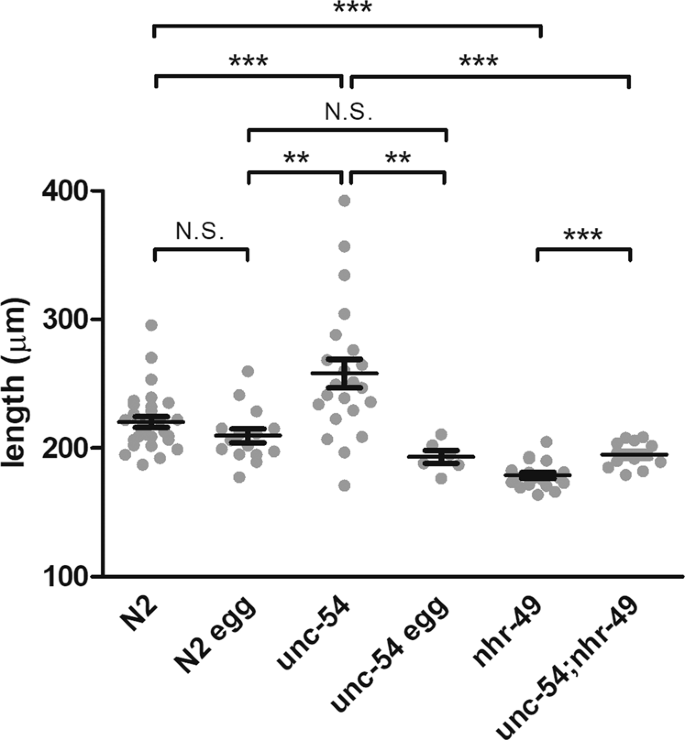
Nutrients obtained from internal hatching ameliorate the developmental effects of violacein. Length of worms measured after 10 days in violacein. N2 indicates wild type mothers placed in OP50-vio, resulting in both externally-laid and internally-hatched larvae. N2 egg indicates eggs that were collected from N2 mothers and directly placed into OP50-vio. Young of unc-54 mutant mothers, which are all internally hatched, grew significantly larger than wildtype. This effect was abolished in isolated eggs from unc-54 that hatched outside the mother. Young of nhr-49 mutant mothers, which produce low levels of oleic acid, do not grow as well even when 100% of the larvae internally hatch (unc-54;nhr-49). Each dot represents average values of different trials. **Indicates p < 0.01, ***p < 0.001.
Oleic acid improves developmental survival in violacein
Previous reports have shown that matriphagy by internally hatched larvae provides the young with a small nutritional benefit in a food-depleted environment15,25. This small amount of food is enough to allow some of the L1 young to develop sufficiently to reach the L2 dauer stage, an alternative developmental stage that allows worms to survive in adverse environments for long periods of time. However, we observed here that matriphagy ameliorates the L2 developmental arrest of a bacterial toxin allowing a portion of the young to reach near adulthood. We wondered then what nutritional component from the mother’s body was responsible for the improved growth in the presence of violacein. Among the possible candidates, we focused on lipids. Fats, both saturated and unsaturated, play integral roles in development, reproduction and aging in the worm27. When compared to the lipids found in bacteria, C. elegans lipids in general tend to contain longer chain fatty acids that reach up to 20 carbon fatty acids27. C. elegans also synthesizes polyunsaturated fatty acids from saturated and monounsaturated fatty acids, and the enzymes responsible for the elongation and desaturation steps have been well-studied (Fig. 5A)28.
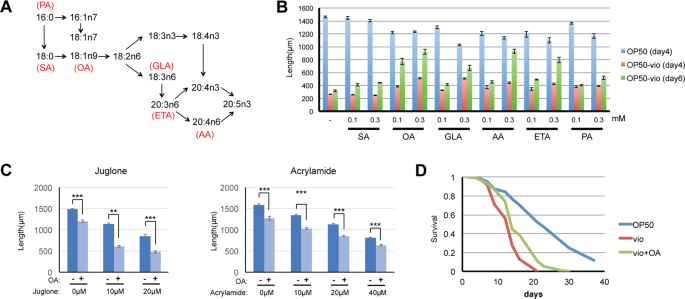
Exogenous supplementation of oleic acid and some PUFAs improve worm growth in OP50-vio. (A) Synthesis pathway of mono-and poly-unsaturated fatty acid from saturated fatty acid in C. elegans (modified from Watts and Browse, 2002). SA = stearic acid, OA = oleic acid, GLA = γ-linoleic acid, AA = arachidonic acid, ETA = eicosatetraenoic acid, PA = palmitoleic acid (B) Worms supplemented with individual fatty acids showed improvements in growth in OP50-vio for 4 days (red) or 6 days (green) at either 0.1 mM or 0.3 mM concentration exogenous fatty acid. Growth in supplemented OP50 is shown in blue. Oleic acid had the most effect, and PUFAs ETA and AA also showed improvement in day 6. All values for fatty acid supplemented samples for OP50-vio 4 days and 6 days have p values <0.001 compared to unsupplemented control (−), except for SA. All values within the same fatty acid are statistically significant (p < 0.001). 30 to 90 worms were tested for each condition. (C) Oleic acid does not mitigate toxicity from the oxidative stressor juglone and neurotoxin acrylamide. Graph shows average of 3 trials. 26 to 43 worms tested for each condition. Statistical significance calculated by Students’ T-test. (D) Oleic acid improves survival of adult worms in OP50-vio. All fatty acids were supplemented at 0.3 mM unless otherwise indicated. Graph show representative results from 3 trials. **Indicates p < 0.01, ***p < 0.001.
To determine whether fatty acids may be a nutritive factor that mitigates the developmental effects of violacein, we tested a number of saturated and unsaturated fatty acids C. elegans can intrinsically synthesize. We analyzed growth of C. elegans larvae simultaneously treated with violacein and the following fatty acids: 18-carbon (SA, 18:0) saturated fat stearic acid, its monounsaturated derivative oleic acid (OA, 18:1n9), and the polyunsaturated fatty acids (PUFA) γ-linoleic acid (GLA, 18:3n6), arachidonic acid (AA, 20:4n6), eicosatetraenoic acid (ETA, 20:4n3) and palmitoleic acid (PA, 16:1n7). We found that after four days of growth in violacein the saturated fatty acid had no effect; however, some of the unsaturated fatty acids could partially attenuate violacein’s toxic effects on growth (Fig. 5B). After 6 days in violacein, untreated worms remain arrested in development, but it was clear now that worms incubated with the monounsaturated oleic acid had a vast improvement in growth. To a lesser extent, omega-6 PUFAs arachidonic acid and eicosatetraenoic acid also had positive effects that became more apparent after day 6. Although oleic acid improves development of worms in violacein, it was previously shown to have toxic effects29. Indeed, when we increased the concentration of oleic acid further, any enhancements were counterbalanced by its negative effects on growth (Fig. S2).
Oleic acid may be alleviating toxicity by either a violacein-specific mechanism or by a general toxicity response. We investigated the latter possibility by testing whether oleic acid can mitigate other forms of toxicity. Juglone is a chemical that induces toxic reactive-oxygen species (ROS). Increasing the concentration of juglone results in higher levels of worm death, but oleic acid treatments did not increase survival (Fig. 5C). Acrylamide is a known neurotoxin, and high levels of acrylamide kill worms30. Oleic acid, though, cannot mitigate the effects of acrylamide either (Fig. 5C). We speculate then that the protective nature of oleic acid against violacein is not functioning via a general stress or toxicity response mechanism.
In addition to improving the worm’s development, we asked whether oleic acid also improves the survival of adults exposed to violacein. In conditions preventing internal hatching by FUDR, the same concentration of oleic acid that improved development also improved the survival of adult worms (Fig. 5D). Thus, oleic acid improves the worm’s ability to overcome the toxic effects of violacein during both larval development and survival.
Oleic acid consumed during matriphagy alleviates the effects of violacein
Although an exogenous application of oleic acid mitigated violacein-induced growth arrest, we wondered whether oleic acid produced in the body of the mother worms and consumed by the young after internal hatching is sufficient to alleviate violacein toxicity. We decided to pursue this question genetically. Mutants of the nhr-49 that encodes a nuclear hormone receptor homologous to the mammalian PPARα show decreased levels of oleic acid31. We wondered, then, if internally hatched young from nhr-49 mutants could still bypass the toxic effects of violacein since their mothers produce less oleic acid. We found that although adult nhr-49 worms were not inherently smaller than N2 adult worms (Fig. S3), larvae born to nhr-49 mutant mothers in violacein were significantly smaller than larvae from wild-type mothers (Fig. 4). To ask whether larvae internally hatched in mothers deficient in oleic acid could grow in violacein, we tested nhr-49;unc-54 double mutant larvae. Compared to how internal hatching visibly improved growth in the presence of violacein (N2 vs unc-54), in the background of the nhr-49 mutation, internal hatching resulted in only a slight mitigation of their growth defects (nhr-49 vs unc-54;nhr-49) (Fig. 4). Hence, we conclude that maternal oleic acid consumed by larvae after internal hatching mitigates violacein toxicity. We note that there is, however, a statistically significant improvement in growth in the internally hatched nhr-49 mutants. This may be due to the physical protection provided by the mother’s cuticle in the early larvae, or due to other nutrients that also contribute to their improved growth in violacein.
Differential toxicity of violacein in species of free-living nematodes
Violacein production is associated with quorum-sensing, a form of pheromonal communication in bacteria. The purple pigment is an excellent indicator that bacterial communities are communicating with one another and preparing to form biofilms. Along with violacein, quorum-sensing induces bacteria to produce hundreds of other secondary metabolites that can have toxic effects to organisms that come in contact with them. Indeed, non-pathogenic bacterial biofilms induce a stress response in C. elegans32, and the pathogenicity of Chromobacterium violaceum towards C. elegans is dependent on quorum-sensing mechanisms33. In light of this, we asked whether violacein toxicity is a conserved mechanism for bacteria to evade bacteriovorous nematode predation. To evaluate this, we examined whether violacein could affect growth in four free-living nematode species, C. briggsae, C. remanei, Pelodera sp., and Pristionchus pacificus. Surprisingly, we found that only two of these species, C. briggsae and Pelodera sp., showed severe developmental arrest similar to C. elegans (Fig. 6A). Larval development in C. remanei, which is most closely related to C. elegans, was only partially compromised, and P. pacificus development was completely unaffected. From this we can conclude that the toxicity of violacein depends on the species of nematode.
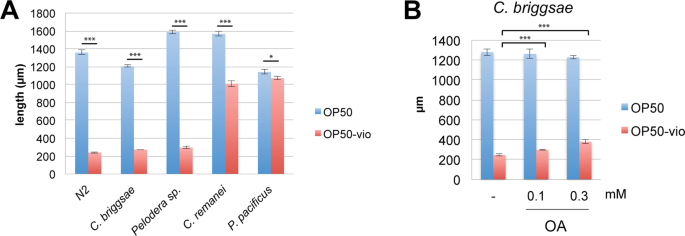
Differential toxicity of violacein in other nematode species. (A) Growth of various nematode species in OP50 and OP50-vio from the L1 larvae stage. Similar to C. elegans, C. briggsae and Pelodera sp. are sensitive to violacein whereas C. remanei development is slightly affected by violacein and P. pacificus are unaffected by violacein. (B) Growth of C. briggsae in violacein is improved by 0.1 mM and 0.3 mM oleic acid (OA) supplementation. ***Indicates p < 0.001, *p < 0.05. Significance determined by Students’ T-test. All graphs show average results of 3 trials.
Mitigation of violacein toxicity by oleic acid is conserved among nematodes
We demonstrated that C. elegans and two other species of nematodes were susceptible to violacein. Could oleic acid also ameliorate violacein toxicity in other nematode species? We examined this question in C. briggsae, a closely related Caenorhabditis member. We found that violacein toxicity to C. briggsae larvae was quite strong, even compared to C. elegans (Fig. 6A,B). Interestingly, exogenous oleic acid could partially ameliorate even these severe effects (Fig. 6B). Thus, we demonstrate here that oleic acid’s beneficial affects against violacein toxicity is conserved in nematodes.
Source: Ecology - nature.com


