Experiment sites and plant materials
Seeds of two fragrant rice cultivars, Meixiangzhan-2 (Lemont × Fengaozhan, bred by Rice research institute of Guangdong academy of agricultural sciences) and Xiangyaxiangzhan (Xiangsimiao126 × Xiangyaruanzhan, bred by Taishan institute of agricultural science), which are well known and widely grown in South China, provided by the College of Agriculture, South China Agricultural University, Guangzhou, China, were used as plant materials in present experiment. A three-year and six-season field experiment was conducted at Experimental Research Farm, College of Agriculture, South China Agricultural University, Zengcheng (23°13′N, 113°81′E, altitude 11 m), China. The experimental site enjoys a subtropical-monsoon climate. The experimental soil was sandy loam consisting of 15.06 g kg−1 organic matter, 1.10 g kg−1 total nitrogen, 53.72 mg kg−1 available nitrogen, 0.83 g kg−1 total phosphorus, 16.37 mg kg−1 available phosphorus, 11.19 mg kg−1 total potassium, and 120.08 mg kg−1 available potassium, and the pH was 6.56. Before sowing, the seeds were soaked in water for 12 h and then were transferred to a constant-temperature (36 °C) incubator for 12 h. Germinated seeds were sown in polyvinyl chloride (PVC) trays for nursery. Fifteen-day-old seedlings were transplanted into paddy field. In early season, fragrant rice cultivars were sown in March, transplanted in April and harvested in July; In late season, fragrant rice cultivars were sown in July, transplanted in August and harvested in November.
Tillage treatment and crop management
Two tillage treatments were applied in present study and the description was as below:
CK: Conventional tillage followed as adopted by local farmers, twice puddling with a rotary cultivator before transplanting in each cropping season;
ZT: Zero tillage, no any tillage was done to the soil in paddy field before transplanting in each cropping season.
The experiment design was a strip plot design with two blocks. Each block was divided into six subplots. The area of each subplot was 90 m2 (3 × 30 m) while the same treatments were applied in the same blocks in each cropping season. The commercial compound fertilizer (YaraMila Fertilizer Company, China) was applied at the same amount of 105 kg·N·ha−1, 105 kg·P2O5·ha−1 and 105 kg·K2O·ha−1 with 60% as basal dose and 40% at tillering. The paddy field was flooded to about 3 cm water depth after the transplanting until the end of the tillering. Then, the water was drained for a week to control the production of infertile tillers and then a water later of 5–7 cm was kept during the grain-filling stage. All other agronomic practices i.e., pest and diseases management, and weed control were the same according the guidelines and standards recommended by the Tang et al.21.
Determination of the 2-AP and proline content in grain at the maturity stage
Ten grams of fresh and mature grain samples were finely ground and then used for measurements of 2-AP via the simultaneous distillation-extraction method (SDE) to make a molar solution, which was then analyzed by a GCMS-QP 2010 Plus instrument (Shimadzu Corporation, Japan). The proline concentration was estimated according to the method described by Du et al.11.
Measurements of yield and yield-related traits
At the maturity stage, the rice grains were harvested from a three-unit sampling area (1.00 m2) in each plot and then threshed by a machine according to methods descript be Du et al.10. The harvested grains dried in the sun, after which they were weighed to determine the grain yield. Twenty hills of rice from each plot were sampled to estimate the average effective panicle number per hill. Six representative hills of plants were then selected to estimate the yield-related traits.
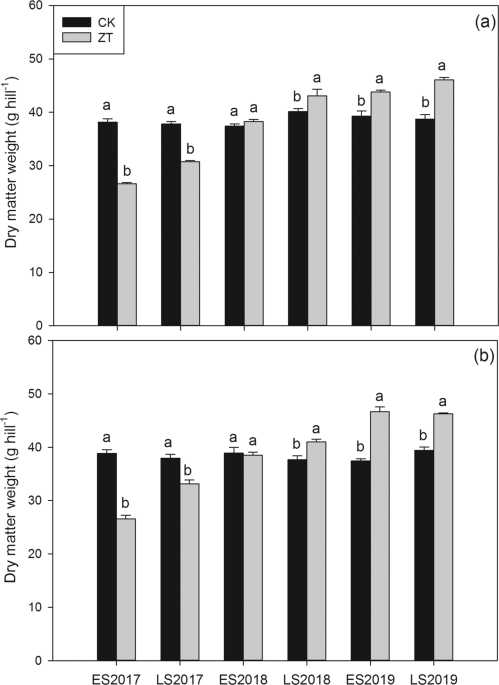
Estimation of biomass accumulation and total nutrient accumulation
At the maturity stage, rice plants of ten representative hill in each treatment were collected and dried out at 80°C for the measurement of dry matter weight and determination of total nitrogen, phosphorus and potassium accumulation in aboveground tissue according to the methods described by Pan et al.22. The rice plants collected were divided into leaf, stems and grain and then oven-dried at 80 °C, weighed, ground into powder for digestion. The digested samples were used to determine the total N content by the Kjeldahl method with a 2300 Kjeltec Analyser Unit (Foss Tecator AB, Swedish). The total P and K concentrations were determined by using the UV-VIS Spectrophotometer UV-2550 (SHIMADZU Corporation) and the Atomic Absorption Spectrophotometer AA-6300C (SHIMADZU Corporation) method, respectively.
Determination of net photosynthetic rate and chlorophyll content
At tillering, heading and grain-filling stage, portable photosynthesis system (LI-6400, LI-COR, USA) was used to determine net photosynthetic rate at 09:00–10:30 a.m. with the following adjustments: photosynthetically active radiation at leaf surface was 1100 and 1200 μmol m−2 s−1, ambient CO2 concentration 380.0–400.0 μmol mol−1. SPAD meter ‘SPAD-502’ (Konica Minolta, Japan) was used for leaf chlorophyll contents.
Determination of soil organic matter and soil microorganism community quantity
Soil samples from 0–20 cm depth were taken for the determination of soil organic matter and microorganisms by the 5-point sampling method at the maturity stage in each season and soil organic matter and soil microorganism community quantity was determinate according the methods described by Du et al.11. The light fraction (LF) and heavy fraction (HF) of soil organic matter were separated and the air-dried soil was homogenized with 30 mL NaI solution (gravity 1.8 g cm−3) in a 100 ml centrifuge tube by shaking on a reciprocating shaker for 60 min at 200 rpm, after which it was centrifuged at 1000 × g for 15 min. The LF, all floating material after centrifugation, was poured into a vacuum filter unit with a 0.45-μm nylon film, and the material retained by the film was washed with 0.01 M CaCl2 and distilled water. The HF remaining in the centrifuge tube was washed three times with ethanol to remove excess NaI, after which it was washed twice with distilled water. Next, the LF and HF were dried at 60 °C for 48 h, and then weighed and ground to pass through a 0.15-mm sieve for the organic determinations. the organic matter in LF and HF were determined by the wet oxidation method with K2Cr2O7 at 170–180 °C. The main soil microorganisms such as bacteria, fungi and actinomycetes were separated and counted by the dilution plate.
Statistical analyses
The data was analyzed using the statistical software ‘Statistix 8.1’ (Analytical Software, Tallahassee, FL, USA), while differences among means were separated by using the least significant difference (LSD) test at the 5% probability level. Graphical representation was performed via Sigma Plot 14.0 (Systat Software Inc., California, USA).
Source: Ecology - nature.com


