Study area
The study was conducted in the Araguari river basin, located in the Brazilian State of Amapá (Fig. 4). Climate in the area is characterized as equatorial monsoon44 with an annual rainfall greater than 2000 mm. The dry season extends between September and November (total monthly rainfall <150 mm), and the wet season from February to April (total monthly rainfall >300 mm)45.
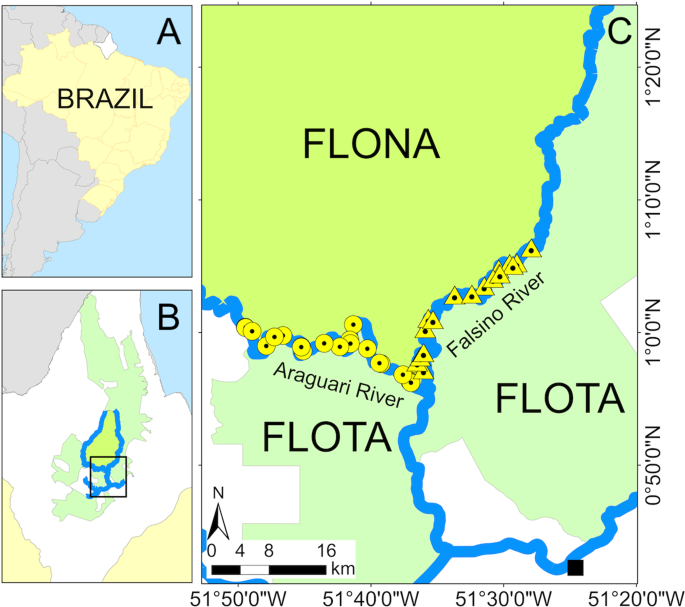
Study area. (A) State of Amapá in Brazil. (B) Location of the study area, FLONA and FLOTA within Amapá. (C) Location of the sampled nest sites along the Araguari (circles) and Falsino (triangles) rivers. Location of the nearest town (Porto Grande) is shown by a black square. Figure produced with ArcGIS 10.4 (https://desktop.arcgis.com/en/).
This study was conducted along a total of 88 km of contiguous segments of Falsino and Araguari rivers, 42 km and 46 km, respectively (Fig. 4). These river segments were located between two sustainable-use protected areas, the National Forest of Amapá (hereafter FLONA), and the Amapá State Forest (hereafter FLOTA). Both segments can be classified as “clear-water”, with a low density of suspended particles46, but they experience markedly different human activity41. The Araguari river is characterized by higher human density (we counted 13 houses in 2017) and more intense boat and fishing activities (including commercial fishing)41,42. In contrast there is no commercial fishing and only 4 houses along the Falsino river.
Turtle nest surveys and sampling
Monthly boat surveys were used to locate nesting areas. Nest searching was conducted between October and December 2017 during the nesting season of Podocnemis unifilis in the region23,25. While navigating along the rivers in a motorized boat at a constant speed (ca. 10 km/h) we performed an extensive search for nesting areas that involved identifying all suitable nesting areas through visually searching river banks, circling islands, stopping to search among boulders and rapids. Nesting areas were identified as areas>5 m2 of exposed substrate sufficiently raised above the river level not to be waterlogged at a depth of 15 cm, a representative depth that females dig when nesting25,34,47. Although we cannot guarantee that all nesting areas were located across 88 km of rivers, we assume that the comprehensive survey effort enables us to provide a robust and representative evaluation of the nest-site substrates.
In all nesting areas, a search for nests was done together with local residents with over 30 years of knowledge on nesting sites25. Searches were conducted by a team of three observers at a standardized speed (mean 0.8, range: 0.2-1.3 km per hour); time spent searching nesting areas ranged from 10 to 97 min depending on the size of the area. To minimize possible observer-related biases, at least one surveyor was constantly present with every search team throughout the entire study period. Logistical limitations hampered our capacity to visit all nesting areas more than once along 88 km of rivers. In order to standardize the number of visits for each nesting area, we included only data from a single visit during the first fortnight of November because this corresponded to the period when both turtle nesting and removal of nests by humans reached their peak during the nesting season30.
Substrate collection and grain size determination
At each turtle nest we collected 200 g of substrate from the nest surface (1 – 3 cm), which was stored in plastic bags labeled with the identifications of the nesting area and the nest. The location of each turtle nest was obtained in the field with a handheld Global Positioning System (GPS) accurate to +2 m. We could not evaluate hatchling success from turtle nests surveyed due to high rates of nest harvest by humans in the study area. For example, in 2017, 42% of the nesting areas along 33 km of the Falsino River (the river segment with the lowest anthropogenic disturbance) were harvested by humans23. Thus, hatchling success in our study region could not be linked directly with nest substrate.
All substrate samples were analyzed at the Laboratório de Análises de Sedimentos do Instituto de Pesquisa Cientificas e Tecnológicas Estado do Amapá – IEPA (Macapá, Brazil). To obtain grain size frequency distribution of substrate samples we used dry sieving, which is a well-established technique48. In the laboratory, all organic material (i.e., vegetation) was removed manually from samples with tweezers. The samples were dried at 50 °C for 24 hours, and were shaken by a mechanical shaker for 10 minutes through a stack of 12 sieves49. The mesh size of the sieves were 2.0 mm, 1.4 mm, 1.0 mm, 0.71 mm, 0.5 mm, 0.355 mm, 0.25 mm, 0.18 mm, 0.125 mm, 0.09 mm, 0.063 mm, and <0.063 mm arranged in a decreasing geometric scale. After shaking, the amount of substrate retained on each sieve was weighed with a precision electronic scale with an accuracy of 0.001 g.
Response variable
To quantify nest removal by humans we identified if each nest was removed or not by humans30, retaining individual nests as the sample unit in the analysis. Human removal of eggs was identified when an open nest was found with a typical depth between 10–15 cm, but lacking eggs. Signs of human activities in the nesting areas, such as footprints, charred wood from fires, campsite or trash were used for corroboration. Nests depredated by wild animals (identified by the presence of broken eggshells and/or partially eaten eggs around the nest, with animal excavation marks were present) though damaged were considered not removed by humans30.
Explanatory variables
We examined a total of 8 (4 environmental, 2 anthropogenic and 2 spatial) variables that were likely to influence nest removal by humans (Table 4, Tables S1–S2). We measured four variables at each nesting area: (1) nesting area type (environmental: either island or bank); (2) river segment (anthropogenic: either Falsino or Araguari), (3) nest density (environmental: expressed as nests per m2); (4) distance to the nearest house (anthropogenic: km). We also recorded two variables at each individual nest: (1) distance to water (environmental: m), and (2) substrate grain sizes (environmental: PC1). We also included geographic coordinates of the nests and the identity of the nesting area to control for possible spatial autocorrelation in the models.
Variables were selected based on previous studies conducted with P. unifilis, as well as our previous knowledge about the study area and species23,25,30,42. For example, nesting area type (island or bank) was found to be an important environmental variable for nest removal by humans at the scale of nesting areas in our study region30. River segment represents different intensities of human activity (i.e., high for Araguari and low for Falsino)41, and it was also highlighted as important predictor for nest removal by humans in previous studies30. Nest density was included as based on classical optimal foraging theory50,51 we would expect relatively increased effort and increased removal where nest density is greater and nests therefore more frequently/easily found. Distance to houses was an important variable to explain nest removal in our study area30, being more important than number of houses within radius of 1 or 5 km for explaining removal between nesting areas. Finally, nest distance to water and proportion of substrate grain size could both affect the detectability by humans. As all nesting sites were located along navigable rivers, and the main transport for riverine community is by boats and canoes, distance of nests from water edge as well as different substrate grain size may result in different detectability and consequent different rates of human removal.
Density of nests was obtained by dividing the total number of nests by the surface nesting site area. This was measured by mapping the surface nesting area using a GPS handheld in situ. Rocks and dense vegetation (non-nesting zones) were excluded from density calculations. Distance to the nearest house was obtained from the centroid point of each nest site to the nearest house (based on GPS fixes obtained in situ at the sampling period). Nest distance to the water’s edge was measured with a measuring tape at the day of the survey. Nesting area size was not included in our analysis as human removal of nests was not influenced by this variable in our study area30.
Substrate type and track visibility
As humans often search for turtle nests by following the tracks of nesting females, previous studies suggest that differences in human removal of turtle nests can be explained by differences in detectability of the tracks in different substrates39. We experimentally tested different substrates to evaluate how the visibility of tracks was affected by different wind speeds. Following the dry sieving, we established three classes of substrate to represent the gradient of fine sand in the 216 nests: 0-10%, 36-46%, and 66-76%. We then selected five nests at random in each class.
In the laboratory samples were placed in tray providing a substrate depth of 6 mm. We drew four tracks in the tray, with size and depth following those recorded from tracks of adult females (Fig. S2). Based on the quantity of substrate collected and size of tray required to maintain a depth of 6 mm it was only possible to draw four tacks. Once drawn, tracks were exposed to different wind speeds (1.5, 3.5 and 5.6 m/s, measured using a digital anemometer) generated by a multispeed electric fan fixed at a constant height and distance from the tray. These speeds represent “light air”, “gentle breeze”, and “moderate breeze” on the Beaufort scale and are typical of those found in the study area during nesting season. The mean wind speed of the central point (0.924722, -51.595833) of our study area is 3.76 m/s according to the information obtained from the “Global Wind Atlas 3.0, a free, web-based application developed, owned and operated by the Technical University of Denmark (DTU). The Global Wind Atlas 3.0 is released in partnership with the World Bank Group, utilizing data provided by Vortex, using funding provided by the Energy Sector Management Assistance Program (ESMAP). For additional information: https://globalwindatlas.info”. The tracks were exposed to wind for five minutes, and after each exposure the number of visible tracks was noted and the tray then reset and the four tracks redrawn.
Data analysis
All statistical analysis and graphics production were undertaken within the R language and environment for statistical computing52. The analysis of grain size samples followed the method of Folk and Ward48 with the packages “rysgran”53 and “soiltexture”54. Principal Component Analysis (PCA) of our seven substrate grain sizes (Table S3) was used to summarize the information of substrate grain size sampled on P. unifilis nests to avoid multicolinearity in subsequent analysis. The PCA was obtained from a scaled and centered data matrix of our seven substrate grain sizes, and axes derived from the squared correlations coefficients with the variables. Based on the Kaiser-Guttman criterion only the first three principal components (with eigenvalues >1) should be interpreted55 (Table S3). Component loadings, summary of principal components after varimax rotation (Table S4), and inspection of correlations revealed that the three axes with eigenvalues >1 (PC1, PC2, and PC3, representing 90% of the variance) were generally represented by proportion of fine sand (negative correlation), medium sand (negative correlation), and gravel (positive correlation), respectively (Fig. S1).
We used Generalized Additive Models (GAMs) to assess the effects of environmental and anthropogenic variables on turtle nests removed by humans. This approach allows the shape of the relationship between the response and the explanatory variables to be determined from the data, rather than following a prescribed functional form56. Thus, we analyzed the response of the nests removed by humans (binary variable), and examined the influence of the explanatory variables (nesting area type, river segment, nest density, distance to the nearest house, distance to water, and scores of PCA first axis related to substrate grain sizes) during the peaks of turtle nesting season and nest removal. A two-step process was adopted with GAMs run (with default settings) using the R package “mgcv”57. Firstly we established whether variables should be include as non-parametric (non-linear) smoothed terms by checking estimated degrees of freedom (EDF) values (typically an EDF value close to or less than 1 suggests linear relationship) and standard diagnostic plots56. Based on results from this preliminary model, we then re-fitted the model including variables as parametric (i.e. linear) terms as appropriate. To control for spatial autocorrelation within and between nesting areas we included the geographic coordinates of the nests as a non-parametric term and the nesting area ID as a random effect (penalized smoothed regression term)56.
Source: Ecology - nature.com


