Bellard, C., Cassey, P. & Blackburn, T. M. Alien species as a driver of recent extinctions. Biol. Lett. 12, 20150623 (2016).
Vilà, M. et al. Explaining the variation in impacts of non-native plants on local-scale species richness: The role of phylogenetic relatedness. Glob. Ecol. Biogeogr. 24, 139–146 (2015).
Gaertner, M. et al. Invasive plants as drivers of regime shifts: Identifying high-priority invaders that alter feedback relationships. Divers. Distrib. 20, 733–744 (2014).
Flory, S. L. & Clay, K. Non-native grass invasion alters native plant composition in experimental communities. Biol. Invasions 12, 1285–1294 (2010).
Potgieter, L. J. et al. Alien plants as mediators of ecosystem services and disservices in urban systems: A global review. Biol. Invasions 19, 3571–3588 (2017).
Ehrenfeld, J. G. Ecosystem consequences of biological invasions. Annu. Rev. Ecol. Evol. Syst. 41, 59–80 (2010).
Liu, Y. et al. Do invasive alien plants benefit more from global environmental change than native plants? Glob. Chang. Biol. 23, 3363–3370 (2017).
Early, R. et al. Global threats from invasive alien species in the twenty-first century and national response capacities. Nat. Commun. 7, 12485 (2016).
Leishman, M. R. & Gallagher, R. V. Will there be a shift to alien-dominated vegetation assemblages under climate change? Divers. Distrib. 21, 848–852 (2015).
Seebens, H. et al. Global trade will accelerate plant invasions in emerging economies under climate change. Glob. Chang. Biol. 21, 4128–4140 (2015).
Jeschke, J. M. & Heger, T. Invasion Biology: Hypotheses and Evidence, https://doi.org/10.1079/9781780647647.0000 (CABI, 2018).
Pickett, S. T. A. et al. Urban ecological systems: Scientific foundations and a decade of progress. J. Environ. Manage. 92, 331–362 (2011).
Cadotte, M. W., Yasui, S. L. E., Livingstone, S. & MacIvor, J. S. Are urban systems beneficial, detrimental, or indifferent for biological invasion? Biol. Invasions 19, 3489–3503 (2017).
Jeschke, J. M. & Starzer, J. Propagule pressure hypothesis. in Invasion Biology: Hypotheses and Evidence (eds. Jeschke, J. M. & Heger, T.) 147–153, https://doi.org/10.1079/9781780647647.0147 (CABI, 2018).
Pyšek, P. et al. Disentangling the role of environmental and human pressures on biological invasions across. Europe. Proc. Natl. Acad. Sci. U. S. A. 107, 12157–12162 (2010).
Aronson, M. F. J., Handel, S. N., La Puma, I. P. & Clemants, S. E. Urbanization promotes non-native woody species and diverse plant assemblages in the New York metropolitan region. Urban Ecosyst. 18, 31–45 (2015).
Elton, C. S. The Ecology of Invasions by Animals and Plants, https://doi.org/10.1007/978-1-4899-7214-9 (Springer, 1958).
Keane, R. M. & Crawley, M. J. Exotic plant invasions and the enemy release hypothesis. Trends Ecol. Evol. 17, 164–170 (2002).
Simberloff, D. & Von Holle, B. Positive interactions of nonindigenous species: Invasional meltdown? Biol. Invasions 1, 21–32 (1999).
McLean, P., Gallien, L., Wilson, J. R., Gaertner, M. & Richardson, D. M. Small urban centres as launching sites for plant invasions in natural areas: insights from South Africa. Biol. Invasions 19, 3541–3555 (2017).
Jeschke, J. M., Debille, S. & Lortie, C. J. Biotic resistance and island susceptibility hypotheses. in Invasion Biology: Hypotheses and Evidence (eds. Jeschke, J. M. & Heger, T.) 60–70, https://doi.org/10.1079/9781780647647.0060 (CABI, 2018).
Rossiter-Rachor, N. A. et al. Invasive Andropogon gayanus (Gamba grass) alters litter decomposition and nitrogen fluxes in an Australian tropical savanna. Sci. Rep. 7, 11705 (2017).
Ehrenfeld, J. G. Effects of exotic plant invasions on soil nutrient cycling processes. Ecosystems 6, 503–523 (2003).
Allison, S. D. & Vitousek, P. M. Rapid nutrient cycling in leaf litter from invasive plants in Hawai’i. Oecologia 141, 612–619 (2004).
Liao, C. et al. Altered ecosystem carbon and nitrogen cycles by plant invasion: A meta-analysis. New Phytol. 177, 706–714 (2008).
Jo, I., Fridley, J. D. & Frank, D. A. More of the same? In situ leaf and root decomposition rates do not vary between 80 native and nonnative deciduous forest species. New Phytol. 209, 115–122 (2016).
Bottollier-Curtet, M., Charcosset, J. Y., Planty-Tabacchi, A. M. & Tabacchi, E. Chemical composition rather than plant geographic origin drives the breakdown of riparian plant litter with changes in associated invertebrate diversity. Plant Soil 390, 265–278 (2015).
Zhu, W. et al. Changes in litter quality induced by nutrient addition alter litter decomposition in an alpine meadow on the Qinghai-Tibet Plateau. Sci. Rep. 6, 34290 (2016).
Aerts, R. Climate, leaf litter chemistry and leaf litter decomposition in terrestrial ecosystems: A triangular relationship. Oikos 79, 439–449 (1997).
Cornwell, W. K. et al. Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol. Lett. 11, 1065–1071 (2008).
González, G. & Seastedt, T. R. Soil fauna and plant litter decomposition in tropical and subalpine forests. Ecology 82, 955–964 (2001).
Ayres, E., Steltzer, H., Berg, S. & Wall, D. H. Soil biota accelerate decomposition in high-elevation forests by specializing in the breakdown of litter produced by the plant species above them. J. Ecol. 97, 901–912 (2009).
Ayres, E. et al. Home-field advantage accelerates leaf litter decomposition in forests. Soil Biol. Biochem. 41, 606–610 (2009).
Yuan, X. et al. Litter decomposition in fenced and grazed grasslands: A test of the home-field advantage hypothesis. Geoderma 354 (2019).
Gholz, H. L., Wedin, D. A., Smitherman, S. M., Harmon, M. E. & Parton, W. J. Long-term dynamics of pine and hardwood litter in contrasting environments: Toward a global model of decomposition. Glob. Chang. Biol. 6, 751–765 (2000).
Ayres, E., Dromph, K. M. & Bardgett, R. D. Do plant species encourage soil biota that specialise in the rapid decomposition of their litter? Soil Biol. Biochem. 38, 183–186 (2006).
Gießelmann, U. C. et al. Lack of home-field advantage in the decomposition of leaf litter in the Atlantic rainforest of Brazil. Appl. Soil Ecol. 49, 5–10 (2011).
Bachega, L. R. et al. Decomposition of Eucalyptus grandis and Acacia mangium leaves and fine roots in tropical conditions did not meet the Home Field Advantage hypothesis. For. Ecol. Manage. 359, 33–43 (2016).
Lin, H. et al. Effect of N addition on home-field advantage of litter decomposition in subtropical forests. For. Ecol. Manage. 398, 216–225 (2017).
Cornelissen, J. H. C. et al. Leaf digestibility and litter decomposability are related in a wide range of subarctic plant species and types. Funct. Ecol. 18, 779–786 (2004).
Tao, J. et al. Traits including leaf dry matter content and leaf pH dominate over forest soil pH as drivers of litter decomposition among 60 species. Funct. Ecol. 33, 1798–1810 (2019).
Pakeman, R. J., Eastwood, A. & Scobie, A. Leaf dry matter content as a predictor of grassland litter decomposition: A test of the ‘mass ratio hypothesis’. Plant Soil 342, 49–57 (2011).
Pérez-Harguindeguy, N. et al. Chemistry and toughness predict leaf litter decomposition rates over a wide spectrum of functional types and taxa in central Argentina. Plant Soil 218, 21–30 (2000).
Cornelissen, J. H. C. et al. Leaf structure and defence control litter decomposition rate across species and life forms in regional floras on two continents. New Phytol. 143, 191–200 (1999).
Liu, G. et al. Specific leaf area predicts dryland litter decomposition via two mechanisms. J. Ecol. 106, 218–229 (2018).
Hättenschwiler, S. & Jørgensen, H. B. Carbon quality rather than stoichiometry controls litter decomposition in a tropical rain forest. J. Ecol. 98, 754–763 (2010).
Hättenschwiler, S., Coq, S., Barantal, S. & Handa, I. T. Leaf traits and decomposition in tropical rainforests: Revisiting some commonly held views and towards a new hypothesis. New Phytol. 189, 950–965 (2011).
Zhang, D., Hui, D., Luo, Y. & Zhou, G. Rates of litter decomposition in terrestrial ecosystems: Global patterns and controlling factors. J. Plant Ecol. 1, 85–93 (2008).
Taylor, B. R., Parkinson, D. & Parsons, W. F. J. Nitrogen and lignin content as predictors of litter decay rates: A microcosm test. Ecology 70, 97–104 (1989).
Meentemeyer, V. Macroclimate and lignin control of litter decomposition rates. Ecology 59, 465–472 (1978).
Frainer, A., Moretti, M. S., Xu, W. & Gessner, M. O. No evidence for leaf-trait dissimilarity effects on litter decomposition, fungal decomposers, and nutrient dynamics. Ecology 96, 550–561 (2015).
Melillo, J. M., Aber, J. D. & Muratore, J. F. Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology 63, 621–626 (1982).
Incerti, G. et al. Faster N release, but not C loss, from leaf litter of invasives compared to native species in mediterranean ecosystems. Front. Plant Sci. 9, 534 (2018).
Hernández, E., Questad, E. J., Meyer, W. M. & Suding, K. N. The effects of nitrogen deposition and invasion on litter fuel quality and decomposition in a Stipa pulchra grassland. J. Arid Environ. 162, 35–44 (2019).
Bumb, I. et al. Traits determining the digestibility-decomposability relationships in species from Mediterranean rangelands. Ann. Bot. 121, 459–469 (2018).
Directorate of Economic and Statistics. Gross State Domestic Product. (Government of India, 2019).
Hui, C., Richardson, D. M. & Visser, V. Ranking of invasive spread through urban green areas in the world’s 100 most populous cities. Biol. Invasions 19, 3527–3539 (2017).
Aragón, R., Montti, L., Ayup, M. M. & Fernández, R. Exotic species as modifiers of ecosystem processes: Litter decomposition in native and invaded secondary forests of NW Argentina. Acta Oecol. 54, 21–28 (2014).
Ashton, I. W., Hyatt, L. A., Howe, K. M., Gurevitch, J. & Lerdau, M. T. Invasive species accelerate decomposition and litter nitrogen loss in a mixed deciduous forest. Ecol. Appl. 15, 1263–1272 (2005).
Anning, A. K., Gyamfi, B. & Effah, A. T. Broussonetia papyrifera controls nutrient return to soil to facilitate its invasion in a tropical forest of Ghana. J. Plant Ecol. 11, 909–918 (2018).
Baruch, Z. & Goldstein, G. Leaf construction cost, nutrient concentration, and net CO2 assimilation of native and invasive species in Hawaii. Oecologia 121, 183–192 (1999).
Harner, M. J. et al. Decomposition of leaf litter from a native tree and an actinorhizal invasive across riparian habitats. Ecol. Appl. 19, 1135–1146 (2009).
Osunkoya, O. O. & Perrett, C. Lantana camara L. (Verbenaceae) invasion effects on soil physicochemical properties. Biol. Fertil. Soils 47, 349–355 (2011).
Blossey, B. & Notzold, R. Evolution of increased competitive ability in invasive nonindigenous plants: A hypothesis. J. Ecol. 83, 887 (1995).
Felker-Quinn, E., Schweitzer, J. A. & Bailey, J. K. Meta-analysis reveals evolution in invasive plant species but little support for Evolution of Increased Competitive Ability (EICA). Ecol. Evol. 3, 739–751 (2013).
Müller-Schärer, H. & Steinger, T. Predicting evolutionary change in invasive, exotic plants and its consequences for plant-herbivore interactions. in Genetics, Evolution and Biological Control (eds. Ehler, L., Sforza, R. & Mateille, T.) 137–162, https://doi.org/10.1079/9780851997353.0137 (CABI, 2004).
Baker, H. G. Characteristics and modes of origin of weeds. In The Genetics of Colonizing Species (eds. Baker, H. G. & Stebbins, G. L.) 147–168 (Academic Press Inc., N.Y., 1965).
Richards, C. L., Bossdorf, O., Muth, N. Z., Gurevitch, J. & Pigliucci, M. Jack of all trades, master of some? On the role of phenotypic plasticity in plant invasions. Ecol. Lett. 9, 981–993 (2006).
Darwin, C. On the Origin of Species by means of Natural Selection. (John Murray, 1860).
Macarthur, R. & Levins, R. The limiting similarity, convergence, and divergence of coexisting species. Am. Nat. 101, 377–385 (1967).
Coûteaux, M. M., Bottner, P. & Berg, B. Litter decomposition, climate and litter quality. Trends Ecol. Evol. 10, 63–66 (1995).
Veen, G. F. C., Sundqvist, M. K. & Wardle, D. A. Environmental factors and traits that drive plant litter decomposition do not determine home-field advantage effects. Funct. Ecol. 29, 981–991 (2015).
Li, Y. et al. Changes in litter quality induced by N deposition alter soil microbial communities. Soil Biol. Biochem. 130, 33–42 (2019).
Bradford, M. A., Berg, B., Maynard, D. S., Wieder, W. R. & Wood, S. A. Understanding the dominant controls on litter decomposition. J. Ecol. 104, 229–238 (2016).
McLeod, M. L. et al. Exotic invasive plants increase productivity, abundance of ammonia-oxidizing bacteria and nitrogen availability in intermountain grasslands. J. Ecol. 104, 994–1002 (2016).
CAB International. Invasive Species Compendium. (2019). Available at, https://www.cabi.org/isc (Accessed: 27th July 2019).
Parker, R. N. A Forest Flora for the Punjab with Hazara and Delhi . (Government Printing, Punjab, 1924).
Khuroo, A. A. et al. Alien flora of India: Taxonomic composition, invasion status and biogeographic affiliations. Biol. Invasions 14, 99–113 (2012).
Doughty, R. W. The Eucalyptus: A Natural and Commercial History of the Gum Tree . (Johns Hopkins University Press, 2000).
Harwood, C. E. Grevillea robusta: An Annotated Bibliography. (International Council for Research in Agroforestry, 1989).
Ghate, V. S. Noteworthy plant invasions in the flora of Western Ghats of Maharashtra. J. Bombay Nat. Hist. Soc 88, 390–394 (1991).
Zanne, A. E. et al. Three keys to the radiation of angiosperms into freezing environments. Nature 506, 89–92 (2014).
Moore, P. D. & Chapman, S. B. Methods in Plant Ecology. (Blackwell Scientific, 1986).
Anderson, J. M. & Ingram, J. S. I. Tropical Soil Biology and Fertility: A Handbook of Methods . Second edition (CABI, 1993).
Van Soest, P. J. & Wine, R. H. Determination of lignin and cellulose in acid detergent fiber with permanganate. J. A.O.A.C. 51, 780–785 (1968).
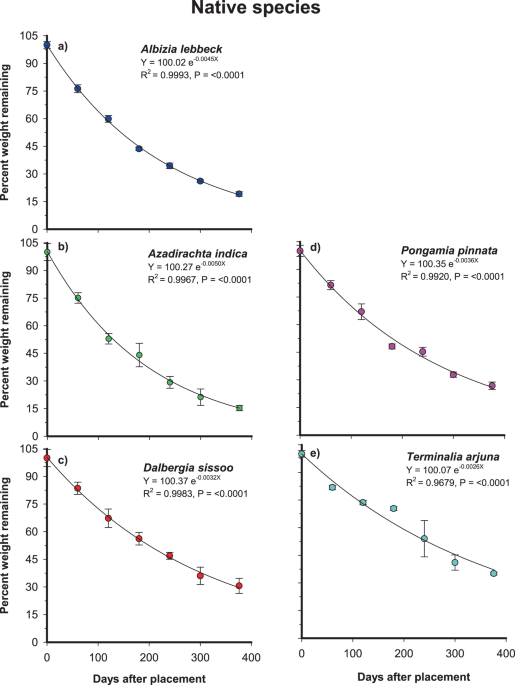
Olson, J. S. Energy storage and the balance of producers and decomposers in ecological systems. Ecology 44, 322–331 (1963).
Peterson, B. G. & Carl, P. PerformanceAnalytics: Econometric tools for performance and risk analysis. Available at, https://CRAN.Rproject.org/package=PerformanceAnalytics (2019).
Kembel, S. W. et al. Picante: R tools for integrating phylogenies and ecology. Bioinformatics 26, 1463–1464 (2010).
R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available at, http://www.Rproject.org/ (2018).
Source: Ecology - nature.com


