Phylogenetic relationship of Alviniconcha marisindica holobionts in the Kairei and the Edmond fields
We cloned and sequenced partial mitochondrial cytochrome c reductase (mtCOI) genes from individuals of both A. marisindica populations. The sequence of the mtCOI gene of kAlv showed 99.4% sequence identity with that of eAlv, indicating that both populations of A. marisindica taxonomically belong to the same species. The phylogenetic tree of the mtCOI gene, including the data obtained from the previous studies [50], also demonstrated that kAlv and eAlv were very closely related (Fig. 2a).
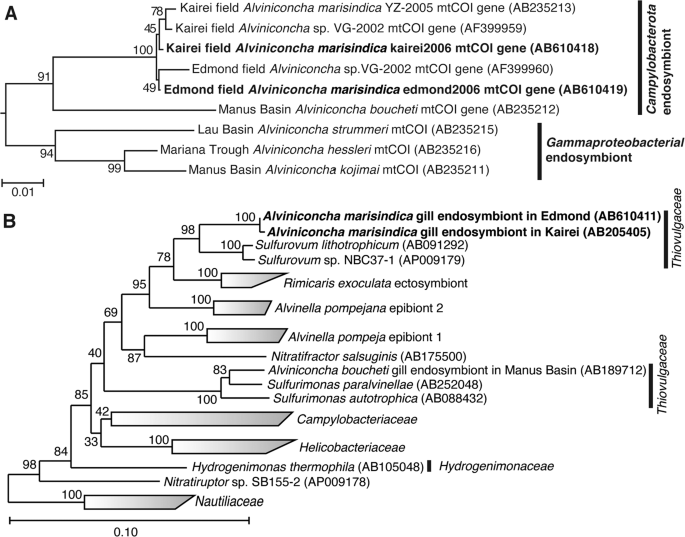
The phylogenetic trees were constructed by the neighbor-joining method using MEGA5 software [45] based on the mtCOI gene sequence (544 base position) (a) and using the ARB software package [44] based on 16S rRNA gene sequences (1402 base position) (b). The mtCOI gene sequences obtained from A. marisindica individuals in this study have been indicated in bold. The mtCOI genes of Ifremeria nautilei from North Fiji Basin (AB235214) and Manus Basin (AB235217) [22] were used as an outgroup for this tree. The 16S rRNA gene sequences obtained from A. marisindica individuals in this study have been indicated in bold. The 16S rRNA gene of Aquifex aeolicus (AJ309733) was used as an outgroup of this tree.
The 16S rRNA genes of endosymbionts of both kAlv and eAlv were cloned and sequenced. The 16S rRNA gene sequences of the kAlv and eAlv endosymbionts determined in this study were identical to the previously reported sequences of them [26] and none of the sequences potentially derived from minor symbiontic bacteria was obtained in the previous and present studies. The sequence identity between the kAlv and eAlv endosymbionts was 99.6%, indicating that the endosymbionts of kAlv and eAlv are taxonomically the same species. The both endosymbionts belong to the genus Sulfurovum within the class Campylobacterota and the most closely related cultivated strain is Sulfurovum sp. NBC37-1, whose genome sequence has previously been determined [19] (Fig. 2b).
These results showed that it may be interpreted that all the molecular and functional properties of both populations in this study are derived from a taxonomically identical holobiont system, the same species of endosymbiont and host animal, although further genome-sequence analyses of both endosymbionts and hosts will clarify the genomic differences between the holobionts of kAlv and eAlv in the future.
Physical and chemical conditions and chemolithotrophic metabolic potentials of in situ habitats of Alviniconcha marisindica in the Kairei and the Edmond fields
During the seafloor observation at the Kairei and Edmond hydrothermal fields, we found several A. marisindica colonies (Fig. 1).
In the Kairei field, the largest colony of A. marisindica (kAlv) was found in the diffusing fluid flow zones at the foot of the Monju chimney site and consisted of several hundred or over a thousand individuals with Chrysomallon squamiferum behind the large population of Rimicaris kairei (shrimp) (Fig. 1a) [26, 28,29,30, 51, 52]. When the temperature of A. marisindica colony water (i.e., the seawater in and around the colony of the A. marisindica population) was measured by a temperature probe equipped with the top of water sampler inlet during the water sampling and sensing (at the positions attaching the shells of individuals), the temperature ranged from 11.9 to 60.0 °C (average 19.3 °C) (Table 1).
To determine the chemical conditions within the kAlv colony, we introduced in situ electrochemical sensors for H2 and H2S concentrations, together with a DO sensor. The H2 concentration of kAlv colony water was found to be 20.1–40.6 μM (average 34.1 μM) (Table 1). The H2S concentration of water in the kAlv colony was found to be 137–211 μM (average 185 μM) (Table 1).
In the Edmond field, we discovered many small colonies (10–20 individuals) of A. marisindica (eAlv) along the cracks with diffusing fluid flows at the foot of the enormous hydrothermal mound (giant shrimp chimney) (Fig. 1b). The temperature of the water in several representative colonies ranged from 6.5 to 38.1 °C (average 26.8 °C) (Table 1). The H2 concentration in water at the eAlv habitats ranged from 0.39 to 0.45 μM (average 0.42 μM) (Table 1), indicating that eAlv inhabits water with much lower H2 concentrations that kAlv. In contrast, the H2S concentration in the eAlv habitats ranged from 109 to 127 μM (average 120 μM), and the DO concentration was found to be 147–202 μM (average 184 μM) (Table 1). In contrast to the H2 concentration, both the kAlv and eAlv would colonize under similar concentrations of H2S and DO.
To determine the H2 and H2S concentrations in both kAlv and eAlv waters, we also used the water samples recovered onboard (Table 1). However, the H2 and/or H2S concentrations in the recovered water samples were strongly reduced probably due to microbial consumption and chemical oxidation during the recovery (Table 1).
Using these physical and chemical properties of the in situ habitats and the bimodal mixing model between each of the endmember hydrothermal fluids and ambient seawater previously described [12], we calculated potential metabolic energy (J/kg water) of H2 oxidization with O2 reduction (aerobic hydrogenotrophy) and H2S oxidization with O2 reduction (aerobic thiotrophy) for free living and/or symbiotic microbial populations in the kAlv and eAlv colonies (Table 2). The calculated energy yield of aerobic hydrogenotrophy (4.4–9.4 J/kg water) using the in situ data of the kAlv colony was considerably lower than the yield based on the mixing model calculation of the kAlv colony (24.1–41.1 J/kg water) (Table 2). In contrast, the energy yield of aerobic thiotrophy in the kAlv colony was similar between the calculations based on the in situ data (39.2–65.0 J/kg water) and the mixing model (31.8–37.9 J/kg water) (Table 2). This was because the measured in situ H2 concentrations were lower than the H2 concentrations simply expected from the bimodal mixing between the endmember fluid and the seawater. Since the diffusing fluids (even high-temperature fluids) had already experienced subseafloor-microbial activities and physical–chemical processes prior to the seafloor discharge [33, 53], the H2 concentrations of the seafloor diffusing fluids were strongly reduced from the initial concentrations during the seafloor mixing process. Even under the in situ H2 concentrations, however, aerobic hydrogenotrophy is thermodynamically energy yielding and is a competitive metabolism with aerobic thiotrophy for the free living and symbiotic microbes in the kAlv population (Table 2).
Similarly, the calculated energy yield of aerobic hydrogenotrophy (0.08–0.1 J/kg water) using the in situ data of the eAlv colonies was found to be considerably lower than the yield (0.3–2.5 J/kg water) based on the mixing model calculation of the eAlv colony, while the energy yield of aerobic thiotrophy was similar between the calculations based on the in situ data (56.1–79.5 J/kg water) and the mixing model (33.7–36.2 J/kg water) (Table 2). The available energy potentials of aerobic hydrogenotrophy in the eAlv colonies were much lower than those in the kAlv colony. Thus, aerobic hydrogenotrophy would be highly disadvantageous for the microbial populations of eAlv colonies, including eAlv endosymbionts, compared with aerobic thiotrophy from the thermodynamic aspect. These thermodynamic estimations suggest that hydrogenotrophic energy metabolism may be the viable option for the kAlv endosymbiont but not eAlv endosymbiont, together with the thiotrophic metabolism.
H2– and sulfide-consumption experiments
Onboard H2– and sulfide-consumption experiments were conducted using both living A. marisindica individuals and dissected whole gill tissues within 12 h of onboard recovery. During the incubation of living kAlv individuals with a supply of H2, the dissolved H2 concentration constantly decreased and the average H2-consumption rate was calculated to be 15.7 ± 8.23 μmol/h/individual (Table 3). In the case of incubations with dissected kAlv gill tissues, the H2-consuming rate was evident but lower than for the living individuals (the average rate was 0.59 ± 0.45 μmol/h/gill) (Table 3). These results clearly indicate that the holobiont of kAlv consumes H2 in the habitats. On the other hand, during the incubations with a supply of sulfide, the average sulfide-consuming rates of kAlv individuals and dissected gill tissues were 20.3 ± 6.4 μmol/h/individual and 2.78 ± 0.92 μmol/h/gill, respectively (Table 3). These rates were slightly higher than the H2-consuming rates.
When the living individuals and dissected gill tissues of eAlv were incubated with H2 and sulfide supplies, the average H2– and sulfide-consuming rates were found to be 6.0 ± 1.6 μmol/h/individual for H2 and 9.8 ± 0.35 μmol/h/individual for sulfide and 0.11 ± 0.21 μmol/h/gill for H2 and 2.1 ± 0.47 μmol/h/gill for sulfide (Table 3). The eAlv individuals showed one-third of the H2-consuming and a half of the sulfide-consuming rates of the kAlv individuals, although the dissected gill tissues of both populations showed similar H2– and sulfide-consuming rates (Table 3). It should be noted that experimental H2 concentration in the eAlv individuals and gill tissue (~100 μM) was much higher than in the natural habitat (0.4 μM, Table 1) and the experimental H2S concentration (~200 μM) was similar to the natural habitat (120 μM, Table 1). Thus, such a high concentration of H2 in the incubation would induce possible hydrogenase expression and activity of the eAlv endosymbionts during incubation, as observed in the previous study of Campylobacterota [35]. However, these results suggest that not only the kAlv holobiont but also the eAlv entity would have the metabolic potential to utilize both H2 and H2S in their natural habitat.
The considerably lower H2– and sulfide-consumption rates of the dissected gill tissues compared with the living individuals were observed in both populations of A. marisindica. One explanation for this may be the potentially limited supply of electron acceptors (for the most part O2 but possibly nitrate also) for H2– and sulfide-oxidation in endosymbiont cells in the dissected gill tissue due to the lack of blood ventilation. Without the blood ventilation of the living individual, electron acceptors may not be sufficiently provided to the intracellularly-immobilized bacterial cells. In addition, the lack of blood ventilation may cause a serious deficiency of endosymbiont viability and metabolic activity in the absence of certain physiological factors from the host. Although we thoroughly washed the shell and body surfaces of the living individuals prior to the experiments, we cannot exclude the inevitable contribution of H2– and/or sulfur-oxidizing microbial populations that adhere to the shell and body surface or in the other tissues to the H2– and sulfide-consumption of the living individual.
Molecular analyses of hydABs, sqr, and soxBC genes of endosymbionts
It is known that H2 oxidation is catalyzed only by a membrane-bound [Ni–Fe]-hydrogenase in the Campylobacterota [10, 34]. Acquired electrons from H2 are transferred to the terminal electron-accepting enzymes, such as polysulfide reductase, cytochrome c oxidase, or denitrification enzymes, potentially via ferredoxin [35]. In the draft genome sequence of the endosymbionts of kAlv, two paralogous hydrogenase genes (hydA1B1 and hydA2B2) were found (unpublished data). The identity in amino acid sequence between both hydrogenases was 49% in HydA and 47% in HydB. On the other hand, sulfur oxidation is likely catalyzed by the Sox multi-enzyme system in Sulfurovum sp. NBC37-1, which is taxonomically closely related to the endosymbiont of A. marisindica [35, 39]. The Sox multi-enzyme system performs the conversion of sulfur compounds to sulfate. Acquired electrons are transferred to cytochrome c oxidase or denitrification enzymes potentially through cytochrome c [35]. The draft genome sequence of the kAlv endosymbiont demonstrated that genes encoding Sox proteins were distributed in two gene clusters (unpublished data). One consisted of soxC–cyc–soxY–soxZ genes, and the other contained soxX–soxY–soxZ–soxA–soxB genes. It is also known that Sqr is involved in the conversion of sulfide to polysulfide in many of the Campylobacterota [35, 54]. During this conversion, electrons are received by quinone and transported to the terminal enzymes. We found a sqr gene encoding type IV sulfide:quinone reductase in the draft genome sequence of the kAlv endosymbiont (unpublished data). These enzymes seem to be basic components for H2– and sulfur-oxidizing energy metabolisms of the endosymbionts of A. marisindica. Therefore, to evaluate the expression level of the transcripts of these functional genes related to hydrogenotrophic and thiotrophic metabolisms, we investigated hydA1B1, hydA2B2, soxC, soxB, and sqr genes from both endosymbionts.
The functional genes from both the endosymbionts of kAlv and eAlv of H2– and sulfur-oxidizing energy metabolisms such as hydABs, sqr, and soxBC genes were successfully amplified using degenerated primers [41, 42] or primers based on the genome sequence from the endosymbiont of kAlv, except for the hydA2B2 gene. We also attempted to amplify the hydA2B2 gene using several primers inferred from the kAlv endosymbiont genome but could not obtain the gene from eAlv endosymbiont (Fig. S3). Thus, we tentatively conclude that the eAlv endosymbiont either has nonhomologous hydA2B2 genes or lacks the hydA2B2 gene. The hydA1B1, sqr, soxB, and soxC gene sequences from both endosymbionts were determined. The amino acid sequence identities of hydA1B1, sqr, soxB, and soxC between the kAlv and eAlv endosymbionts were 99.8% (1 amino acid difference per 149 amino acids on HydB), 99.5% (2 amino acid differences per 247 amino acids), 99.9% (1 amino acid difference per 257 amino acids), and 100% (0 amino acid differences per 231 amino acids), respectively.
In addition to the 16S rRNA gene sequence comparison, these results reinforce the idea that both endosymbionts are equipped with almost identical gene repertoires for H2– and sulfur-oxidizing energy metabolisms, except for the hydrogenase paralogs. In other words, the different metabolic behaviors between the holobionts of kAlv and eAlv are derived primarily from regulation at the transcription, expression, and modification levels, responding to the environmental conditions rather than from the genome and genetic structures. Phylogenetic analysis also showed that these genes from the endosymbionts are closely related with those of Sulfurovum species, sharing more than 97% 16S rRNA identity with the endosymbionts of both kAlv and eAlv (Figs. S4–S7). Based on the phylogenetic tree of HydB, it was suggested that the H2-oxidization of Campylobacterota could be attributed to the products of hydA1B1 genes (Fig. S3), and we expected that the hydA2B2 gene products were not responsible for the H2-oxidization of the kAlv holobiont. However, as the functions of the hydA2B2 gene products are unknown, we conducted analyses using both hydA1B1 and hydA2B2 genes.
In addition, to normalize the comparative expression abundance of the functional genes between both kAlv and eAlv endosymbionts, we used gyrB and gap genes, which encode the DNA gyrase B subunit and glyceraldehyde 3-phosphate dehydrogenase, respectively. These genes are widely used as internal controls for the functional gene expression of bacteria and humans [55,56,57,58]. The amplified fragments of gyrB (695 bp) and gap (620 bp) shared 99.6% (three-bases mismatch) and 99.8% (one-base mismatch), respectively, with the endosymbionts.
Estimation of cellular expression of functional gene transcripts in endosymbionts
To evaluate the cellular expression of the functional genes involved in the H2– and sulfur-oxidizing energy metabolisms, we conducted synthesis of cDNAs derived from the transcripts of 16S rRNA, hydABs, sqr, soxB, soxC and reference genes in the endosymbionts from individuals of both kAlv and eAlv. Total RNA assemblages were extracted from the gill tissues of both populations and RT of the total RNAs were conducted. In these procedures, we confirmed that all the double-stranded DNA was completely removed before the RT step by DNaseI digestion (Fig. 3a, b). Then, RT-PCR was performed with the synthesized cDNA assemblages using the primer sets specific to each of the target genes of 16S rRNA, hydAB, sqr, soxB, and soxC genes. The agarose gel electrophoresis of the RT-PCR products demonstrated that the amplifications of 16S rRNA, sqr, and soxB cDNAs were observed in both the kAlv and eAlv gills while the amplification of the hydA1B1 gene was observed only in the kAlv gill under the experimental conditions (Fig. 3a, b).
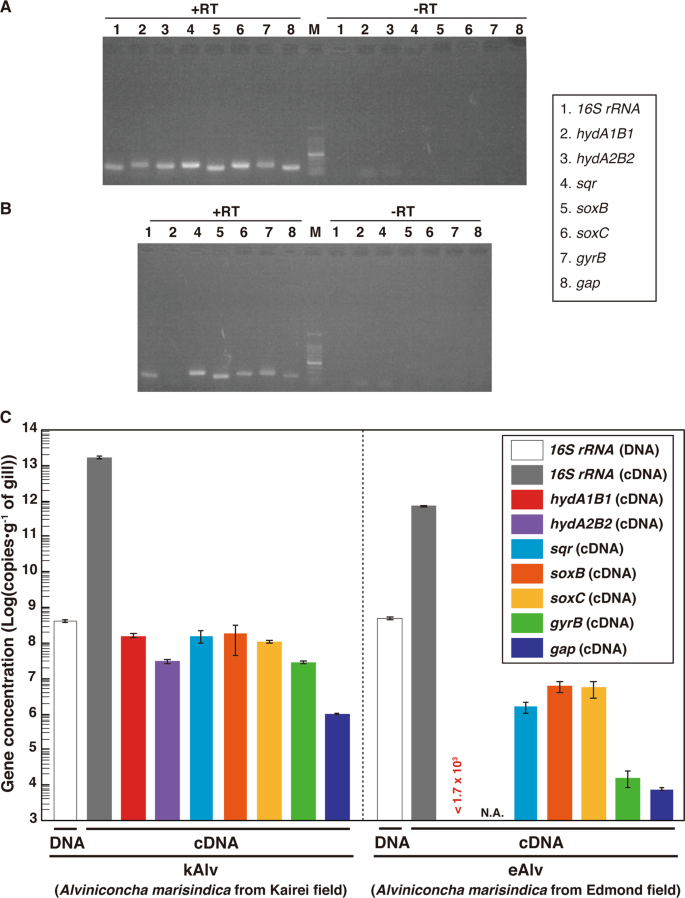
a Agarose gel electrophoresis of PCR products for 16S rRNA, hydABs, sqr, soxBC, gyrB, and gap genes with or without RT of total RNA extracts from kAlv gills. b Agarose gel electrophoresis of PCR products for 16S rRNA, hydA1B1, sqr, soxBC, gyrB, and gap genes with or without RT of total RNA extracts from eAlv gills. c Comparative abundances of 16S rRNA gene in the DNA extracts from kAlv and eAlv gills by quantitative PCR analysis and of hydA1B1, hydA2B2, sqr, soxB, gyrB, and gap gene transcripts in the cDNA assemblages from kAlv and eAlv gills by quantitative RT-PCR analyses. Quantity of transcript (<1.7 × 103 copies/g) of hydA1B1 gene in eAlv holobiont was lower than the detection limit by qPCR analyses. NA indicates not analyzed because the hydA2B2 gene was not detected by PCR in the eAlv endosymbiont.
To further estimate the expression levels of functional genes, we performed qPCR analyses. We confirmed the sensitivity and confidence of quantification in our method by using the clones constructed in the cloning step (Fig. S8). First, we conducted qPCR analysis for the 16S rRNA gene of the endosymbiont using the total DNA extracts from the kAlv and eAlv gills. The estimated quantity of 16S rRNA was 4.5 × 108 and 5.4 × 108 copies/g for kAlv and eAlv, respectively (Fig. 3c). This result indicated that the endosymbiont cell number in 1 g of gill tissue was similar between the kAlv and eAlv individuals. However, when qPCR analysis of endosymbiont 16S rRNA was conducted using the cDNA assemblages from the kAlv and eAlv gills, it was found that the estimated number of 16S rRNA was 22 times higher in the kAlv (1.71 × 1013 copies/g) than in the eAlv (7.66 × 1011 copies/g). This result suggested that the protein synthesis activity of the kAlv endosymbiont was much higher than that of the eAlv endosymbiont. In response to the different abundances of transcript 16S rRNA, we performed qPCR analyses of the gyrB and gap gene transcripts, which were used as references for the expression levels of constitutional genes. The results confirmed that the abundances of gyrB and gap transcripts in the kAlv endosymbiont (3.11 × 107 and 9.94 × 105 copies/g, respectively) were 1870 times greater in gyrB and 130 times greater in gap than those in the eAlv endosymbiont (1.66 × 104 and 7.73 × 103 copies/g, respectively). These results indicate that the expression abundances of constitutional genes related with cellular replication, translation, and central metabolism in the endosymbionts are greater in the kAlv than in the eAlv.
In a similar manner, the RT-qPCR indicated that all functional genes analyzed in this study were more abundantly expressed in the kAlv than in the eAlv (Fig. 3c). These results strongly suggest that the kAlv endosymbiont would be functionally more active than the eAlv endosymbiont. The largest difference of expression between the kAlv and eAlv gills was found in the hydA1B1 gene. The number of hydA1B1 gene transcripts for the kAlv was estimated to be 1.60 × 108 copies/g, while the number for eAlv was below the detection limit (<1.7 × 103 copies/g) (Fig. 3c). However, the estimated abundances of sqr, soxB, and soxC gene transcripts in the kAlv gill were 1.61 × 108, 2.00 × 108, and 1.18 × 108 copies/g, respectively, which were 20–100 times greater than that of the eAlv gill (1.74 × 106, 6.81 × 106, and 6.04 × 106 copies/g for the sqr, soxB, and soxC gene transcripts, respectively) (Fig. 3c). In comparison with various functional genes in the endosymbionts, these results demonstrated that the functional genes for H2– and sulfur-oxidizing energy metabolisms were expressed at similar levels in the kAlv gill, while the functional genes for sulfur-oxidizing energy metabolism were more abundantly expressed than for H2 oxidation in the eAlv gill. In addition, the expression abundance ratios of hydrogenase and sulfur-oxidizing genes against the gap gene were 47.1 (for hydA1B1), 47.5 (for sqr), 59.0 (for soxB), and 34.9 (for soxC) in the kAlv endosymbiont, and <0.021 (hydA1B1), 20.0 (sqr), 78.0 (soxB), and 69.2 (soxC) in the eAlv endosymbiont. These values were similar between the kAlv and eAlv endosymbionts except for the expression of hydA1B1 gene, suggesting the possible functional relevance between energy and carbon metabolisms such as glycogenesis and glycolysis.
Although the hydA2B2 gene was only found in the kAlv endosymbiont, the estimated abundance of the transcribed gene in the gill tissue was 3.26 × 107 copies/g, 4.9 times lower than that of hydA1B1 gene (Fig. 3c). It seems likely, therefore, that the H2 oxidation of the endosymbiont is primarily catalyzed by products of the hydA1B1 gene, although the function of hydA2B2 is still uncertain.
In the kAlv endosymbiont, the results of functional gene expressions seem to be consistent with the results of the H2– and sulfide-consumption experiments of the living individuals. The kAlv holobiont showed higher H2– and sulfide-consuming activities than the eAlv holobiont (Table 3) and showed a similar level of activity for both H2– and sulfide-consumption (Table 3). In contrast, although the eAlv holobiont showed slightly lower H2-consuming activity than sulfide-consuming activity (Table 3), the expression of hydA1B1 genes in the eAlv endosymbiont was below detection limit (<1.7 × 103 copies/g gill) (Fig. 3c). As discussed above, this difference between the onboard consuming experiment and the in situ transcript analysis would be derived from the possible experimental induction of hydrogenase expression and activity by the relatively high H2 concentration used for the onboard consuming experiment of eAlv holobiont. It seems likely therefore that the dual energy metabolism configuration of A. marisindica holobionts may be regulated by the expression of related functional genes responding to the physical and environmental H2 conditions of their habitats.
FISH analyses of hydA1B1 and soxB gene transcripts in gill tissue
To identify the localization of the functional gene transcripts in the gills of both populations, we designed probes to target the hydA1B1 and soxB gene transcripts that were likely involved in the H2– and sulfur-oxidation metabolisms, respectively, and then conducted FISH analysis using the individuals fixed in situ. Consistent with the distribution pattern of endosymbionts shown in the previous study of Alviniconcha [59], signals for the 16S rRNA of Campylobacterota endosymbionts in both populations were detected throughout the apical side of the gill epithelium (Fig. 4). Similarly, signals for the hydA1B1 and soxB gene transcripts were detected at the apical side of the gill epithelium in both the kAlv and eAlv, and mostly overlapped with the signals for the 16S rRNA (Fig. 4). When a sense probe was used as a control, no positive signal was observed (Fig. S9 in Supplemental materials). These results verified that both hydA1B1 and soxB expression occurred not only in the kAlv endosymbiont cells but also the eAlv ones. The FISH results seem to contradict with the qPCR results indicating that the abundance of hydA1B1 transcript in the eAlv endosymbiont was estimated as below detection limit (<1.7 × 103 copies/g gill) and was much lower than that of the kAlv endosymbiont (Fig. 3c). Although other interpretations cannot be excluded, a possible explanation is that the given incubation time of catalyzed FISH in this study may have resulted in excess amplification of initial signals derived from the probes bound with the target functional gene transcripts and may not properly represent the cellular abundances of hydA1B1 and soxB gene transcripts in the kAlv and eAlv endosymbionts. However, it is evident that the hydA1B1 and soxB genes are co-expressed in the identical Campylobacterota endosymbiont cells in the Alviniconcha gill tissues.
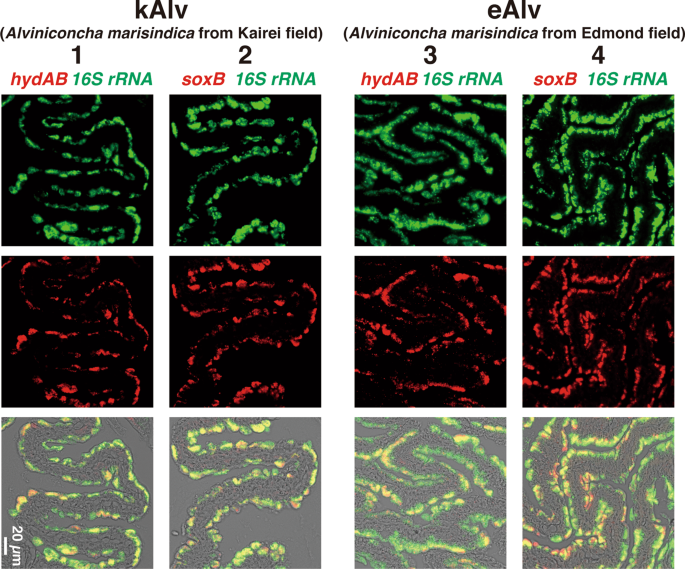
Each column shows the same section image taken by different excitation wavelengths of light. Left two columns (1 and 2) and right two columns (3 and 4) show the results of kAlv and eAlv gills, respectively. Green signals in the top panels are derived from 16S rRNA, and red signals in the middle panels are from hydAB gene transcripts using antisense probe (columns 1 and 3) and soxB gene transcripts using antisense probe (columns 2 and 4). The bottom panels show the synthesized fluorescence of 16S rRNA and each functional gene transcript.
Enzymatic activity analysis in crude gill extract
To estimate the enzymatic basis of H2– and sulfur-oxidizing energy metabolisms in the holobionts of kAlv and eAlv, we determined specific activities of hydrogenase and Sox enzymes in the crude extracts of gill tissues from both kAlv and eAlv individuals. We also attempted to determine the specific activity of Sqr in the gill crude extracts from both populations, but it was difficult to detect the Sqr activity due to the high concentration of various types of proteins in the crude extracts, which interfered with the spectroscopic measurement of the reduced form of quinone.
The specific activity of hydrogenase in the kAlv gill crude extract was found to be 14.1, 51.8, and 94.4 U/g at 25 °C, 45 °C, and 65 °C, respectively (Fig. 5a). In contrast, the specific activity of hydrogenase in the eAlv gill crude extract was 0.025, 0.16, and 1.22 U/g at 25 °C, 45 °C and 65 °C, respectively. It was interesting that the hydrogenase activities of A. marisindica endosymbionts were thermophilic despite the average in situ temperatures of their habitats (20–30 °C) (Table 3). The comparison of hydrogenase activities in the gills clearly indicated that the kAlv holobiont had much greater activity (>100 times) than the eAlv holobiont. The difference of hydrogenase activity between the kAlv and eAlv holobionts was consistent with the difference in hydA1B1 gene expression level between them (Figs. 3 and 5a). Although the living kAlv individuals or their dissected gills showed only three to six times higher H2-consumption activity than living eAlv individuals or their dissected gills under the H2-supplemented incubation condition (Table 3), the gill tissues of freshly sampled individuals from the habitats showed drastic differences in expression and activity of the H2-oxidizing enzyme between the kAlv and eAlv holobionts. The differences observed in H2-consuming experiments using individuals or gill may be due to the initial hydrogenase abundance in the endosymbiont shown in this enzyme assay using crude extract.

The activities of both enzymes were measured at 25 °C, 45 °C, and 65 °C. Red and blue bars indicate the specific activities of hydrogenase (a) and Sox enzymes (b) in crude extracts of gills from kAlv and eAlv, respectively.
On the other hand, the specific activities of Sox enzymes in the kAlv gill crude extracts were 0.140, 0.362, and 0.299 U/g at 25 °C, 45 °C, and 65 °C, respectively, while those in the eAlv gill crude extract were found to be 0.404, 0.500, and 0.362 U/g at 25 °C, 45 °C, and 65 °C, respectively (Fig. 5b). The Sox enzymes from both the kAlv and the eAlv gills showed the highest specific activity at 45 °C, despite the highest activity of hydrogenase observed at 65 °C. This result may represent the substantial enzymatic property of Sox enzymes of endosymbionts, while it is probably due to an analytical artifact because the cytochrome c used in the Sox enzyme activity assay is obtained from bovine serum (Sigma) and thus becomes unstable at 65 °C. The comparison of Sox enzyme activities in the gills indicates that both the kAlv and the eAlv holobionts had similar activities. This result slightly contradicts the comparative results of sulfide-consumption activities of living individuals and expression abundance of soxB gene between the kAlv and eAlv holobionts, in which the sulfide-consumption activity of living individuals and the soxB gene expression were greater in the kAlv holobiont than in the eAlv holobiont (Table 3 and Fig. 3b).
Since the specific activities of hydrogenase and Sox enzymes are usually proportional to the abundance of enzymes in the extract solutions and are substantially coupled with the net energy yield, the difference seen in the enzyme activities in the gills may be derived from the different cellular expression of hydrogenase and Sox enzymes potentially responding to the environmental H2 concentrations in their habitats.
Conclusive remarks and future perspectives
In this study, the chemosynthetic holobionts of two populations of A. marisindica in the CIR deep-sea hydrothermal fields were investigated to justify the hypothesized occurrence of dual energy metabolisms (H2– and sulfur-oxidation). Based on the phylogenetic analysis of representative genes from the endosymbionts and hosts, the two populations (kAlv and eAlv) had approximately identical holobiont systems that consisted of the same endosymbiotic and host species (Fig. 2a, b). The polyphasic physiological and molecular analyses clearly demonstrated that the holobionts of the two populations had metabolic potentials of both H2– and sulfur-oxidation (Fig. 3 and Table 3) but showed different configurations and operations of the dual energy metabolisms. The results strongly suggest that the kAlv holobiont adopts H2 oxidation as its primary energy metabolism as much as, or possibly more than, sulfur oxidation (Figs. 3c and 5a), while the eAlv holobiont was, for the most part, dependent on sulfur oxidation for chemosynthetic production (Figs. 3c and 5a). In the previous studies of Alviniconcha spp. in the ESLC [14, 16], it was suggested that the energy metabolisms of holobionts were controlled by the niche separation of different genetic and metabolic holobiont types (different couples of host and symbiont species) responding to the physical and environmental H2 conditions of the habitats. However, in the CIR A. marisindica, it is shown that the energy metabolisms of holobionts are regulated by the expression and function of dual energy metabolic genes and enzymes in the same holobiont type (the same couple of host and symbiont species). However, due to the limited opportunities for, and numbers of, dive observations, onboard experiments, and samples, statistical verification of different expression and function patterns in dual energy metabolisms was not fully conducted. In addition, as shown in the previous study of mussels in the MAR deep-sea hydrothermal fields [13], the carbon fixation and transport of holobionts associated with energy metabolisms should be clarified. These experiments will be our foci in future studies.
What would drive different configurations and operations of dual energy metabolisms in the taxonomically near-identical holobiont type of A. marisindica? At present, it seems very likely that the in situ H2 concentration of the habitat serves as one of the key factors. The kAlv holobiont dwells in habitats where the H2 concentration is around 100-fold higher than that of the eAlv holobiont (Table 1). If the environmental abundance of H2 can induce the relatively rapid change of configuration and operation of dual energy metabolisms, the hypothetical habitat exchange experiment between the kAlv and eAlv holobionts would result in the transformation of hydrogeno- and thiotrophic eAlv and thiotrophic kAlv holobionts from the native phenotypes. If other substantial factor(s) other than H2 (e.g., strict genotype selection and acquisition of endosymbiont at the initial holobiont formation and intergenerations of host-symbiont genetic interaction responding to the environmental conditions) can control the configuration and operation of dual energy metabolisms, the habitat exchange experiment would not affect the expression of new phenotypes. Not only habitat exchange experiments but also onboard rearing experiments under different H2 conditions will provide important insights into understanding the energy and carbon metabolisms of chemosynthetic symbioses in deep-sea invertebrates. For onboard rearing experiments in the future, the effect of in situ hydrostatic pressure may be considered, although it was revealed that the in situ hydrostatic pressure did not affect the specific activities of energy metabolisms in the case of the symbiotic microbial community of Shinkaia crosnieri (squat lobster) [39].
The H2-depdendent chemosynthetic symbiosis of deep-sea invertebrates was first demonstrated in a deep-sea mussel, B. puteoserpentis, living in the Logatchev hydrothermal field of MAR by Petersen et al. [13]. Based on the FISH and immunohistochemistry analyses and the radio-isotope-labeled bicarbonate tracer experiments, Petersen et al. [13] pointed out that hydrogenotrophy may serve as primary energy metabolism competitively with thiotrophy in the chemosynthetic holobiont. In addition, it has been reported that some chemosynthetic holobionts may use H2 as an energy source because the genes encoding hydrogenase, and even the expression of the genes, were found in the symbiont genomes and transcriptomes [14, 16,17,18,19,20]. However, it has been also shown that these holobionts have molecular and physiological potentials of other energy metabolisms (sulfur and/or methane oxidation). Actually, Mitchell et al., demonstrated that the endosymbionts of tubeworm, Riftia pachyptila, which possessed genes for hydrogenases did not utilized hydrogen but utilized sulfide as a major electron donor [60]. Thus, the physiological demand and metabolic response to the target energy source in the chemosynthetic holobiont should be addressed at multiple levels, including the environmental conditions of habitats, the transcription level of related genes, the expression level of related enzymes and the physiological level of individuals. In this context, we provide multiple lines of evidence that a deep-sea gastropod, A. marisindica, and its endosymbiont in the Kairei field utilize H2 as the primary energy source as well as H2S (sulfide).
Source: Ecology - nature.com


