Site description
The Spruce and Peatland Responses Under Changing Environments (SPRUCE) experimental site (http://mnspruce.ornl.gov/), S1 Bog, is an 8.1 ha peatland in north-central Minnesota, USA within the US Forest Service Marcell Experimental Forest (N 47°30.476′; W 93°27.162′). Since the 1960s, extensive scientific investigations have been done at this site and include in-depth descriptions of its physicochemical and biotic characteristics14,22,23,24,25,26,29,35. This precipitation-fed, ombrotrophic bog has a perched water table with an average pH of 4.1 at the surface which increases with depth to roughly 5.1 at 2 m depth. The overstory vegetation is primarily dominated by Picea mariana (black spruce) and secondarily by Larix laricina (larch), while the understory is composed of low ericaceous shrubs, such as Rhododendron groenlandicum (Labrador tea) and Chamaedaphne calyculata (leatherleaf), and herbaceous perennials, such as Maianthemum trifolium (three-leaved Solomon’s seal) and Eriophorum vaginatum (cotton grass). The bog surface is characterized by hummock and hollow microtopography, with a typical relief of 10 to 30 cm between the tops of the hummocks and the hollows. Sphagnum magellanicum generally colonizes the hummocks, while S. angustifolium and S. fallax cover the hollows. The belowground peat profile and geochemistry are described in ref. 26.
Whole-ecosystem warming and elevated atmospheric [CO2]
The SPRUCE project uses a regression-based experimental design that warms the vegetation and peatland soil profile to 3 m depth within ten, 12-m-diameter enclosures to five target temperature differentials (+0, +2.25, +4.5, +6.75, and +9 °C), with duplicate enclosures subjected to ambient and ~ +500 p.p.m.v. atmospheric CO2 concentrations (eCO2; Supplementary Fig. 1a). Whole-ecosystem warming (WEW) is achieved within open-topped enclosures (7 m tall by 12.8 m in diameter) by combining air and belowground warming. Air is warmed with propane heaters, whereas belowground warming is attained using low-wattage, 3-m deep, belowground concentric rings of heaters11. The open-top enclosure design allows for surface air warming and enhancement of atmospheric CO2, while subsurface corrals hydrologically isolate each experimental enclosure and allow for changes in water-table level associated with climate manipulation to occur.
Whole-ecosystem warming was initiated 12 August 2015, following 14 months of deep-peat heating (DPH). During the DPH phase of this experiment, deep-soil temperature targets were successfully maintained throughout the year following a gradual treatment equilibration period (~3 months); however, the lack of air warming resulted in reduced temperature separation among treatments at the surface11. After the introduction of air warming (which signaled the start of WEW), we attained 9 °C temperature separation and differentials across treatment enclosures from the tops of the trees to peat depths of at least 2 m. Temperature differentials have largely been maintained thought the WEW period, with some variation observed in surficial peat zones due to rain and snow events11. Finally, eCO2 was introduced in a subset of the enclosures on 15 June 2016, completing the full set of experimental climatic manipulations planned by the SPRUCE project (Supplementary Fig. 1b). In these enclosures, ambient atmospheric CO2 concentrations were on average elevated by +500 p.p.m.v. using pure CO2 from a fossil source (i.e., 14C-free CO2). The mixture of local ambient air (+0 to 9‰) with pure CO2 yielded eCO2 chambers having typical values of −520 to 540‰ on the Δ14C scale. Due to the compounding effect of the treatment, we include all 10 enclosures in most analyses and explicitly explore eCO2 in some cases.
CH4 and CO2 production
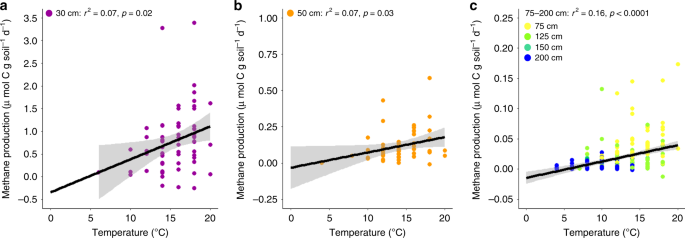
Following the same protocol that was used throughout the DPH experimental phase22, intact soil cores were collected from multiple depths within each experimental enclosure to discern how rates of CH4 and CO2 production, as well as CO2:CH4 ratios, varied with climate treatment and depth. Sampling events were conducted 1–4 times per year during the growing season, and over the course of 4 years (2015–2018) throughout WEW. In 2015, soil cores were collected from 20 to 30, 50 to 75, 100 to 125, 125 to 150, and 175 to 200 cm depth increments (depth increments are denoted with the lower end of their ranges in figures). We used the same sampling protocol from 2016 to 2018, but collected soil cores at 40–50 cm instead of 125–150 cm to better capture variation in surficial peat horizons. All depths were measured relative to the surface of the hollows. To prevent compression of surface peat samples, a serrated knife was used to collect a 10 cm diameter core from the hollow surface to ~20 cm within the peat profile. A 5-cm diameter Russian corer was subsequently used to extract the remaining samples up to 2 m deep. Soil cores were immediately flushed with nitrogen (N2) in the field to minimize exposure to aerobic conditions. In addition, porewater samples were anaerobically collected from within each enclosure using 1.25 cm diameter PVC piezometers installed at corresponding depth increments (25, 50, 75, 100, 150, and 200 cm below the hollow surface) and a peristaltic pump. Both soil cores and porewater were stored on ice and shipped overnight to the University of Oregon (UO) or Chapman University (CU). We began to observe water-table drawdowns (~30–50 cm below the hollow surface) in 2016 as a result of increased temperatures in experimentally manipulated enclosures11. We focus here only on depth increments that were anaerobic at the time of sampling.
In the laboratory, soil samples were incubated within 1 °C of in-situ temperatures within 48 h of field collection and anaerobic incubations commenced the following day. This rapid turnaround time was intended to generate depth-specific CH4 and CO2 production rates that were as representative of in-situ conditions as possible. Samples were slurried with a 1:1 mixture of peat and porewater collected from the same enclosure and depth in a glove box filled with a N2 atmosphere (<5% H2 in the presence of a palladium catalyst) to maintain anaerobic conditions. Sample bottles were then flushed with N2 for 15 min to begin the incubation. Headspace samples were analyzed over the course of 8 days (days 2, 4, 6, and 8) for CH4 and CO2 simultaneously using an SRI gas chromatograph equipped with a methanizer and flame ionization detector. Total CH4 and CO2 were calculated using Henry’s Law and adjusting for solubility, temperature, and pH36. Methane and CO2 production rates were calculated using the linear accumulation (r2 ≥ 0.83 in all cases) of gasses through time.
Porewater chemistry and isotopic composition
During WEW, porewater samples were collected 1–4 times per year during the growing season for analysis of di-hydrogen (H2), acetate, CH4, CO2, and DI14C using 1.25-cm diameter PVC piezometers permanently installed at 25, 50, 75, 100, 150, and 200 cm below the hollow surface within each experimental enclosure. The 1.25-cm diameter opening was parallel to the sampling depth with a screen covering to prevent solid intrusion. Piezometers were pumped out and allowed to passively refill for 24 h prior to sampling. Given the length of the piezometers and small cross-sectional area (1.25 cm diameter), little exchange with the atmosphere was expected over 24 h. A peristaltic pump and flexible sections of silicon tubing were used to collect porewater samples from piezometers, while surface water samples were collected using perforated stainless-steel tubes that were inserted into the peat to 10 cm or the top of the water table, whichever was shallowest.
Di-hydrogen samples were collected four times during the growing season in 2016. Immediately following collection, porewater was stored in pre-evacuated glass vials sealed with butyl stoppers, and phosphoric acid was added to each sample to preserve for shipment to the UO. At the UO, headspace samples were analyzed for H2 using a Peak Performer gas chromatograph with a reducing compound photometer. The level of detection for H2 was 1 ppm mL−1.
Acetate samples were collected 1–4 times during the growing season in 2015, 2016, and 2018. Samples were filtered through a Whatman Grade GF/D glass microfiber pre-filter and a Whatman Grade GF/F glass microfiber filter, stored in 30 mL polycarbonate bottles, and immediately frozen on dry ice. Samples were shipped to CU for analysis. At CU, samples were further filtered through a Whatman 0.2-µm filter in the laboratory. Acetate concentrations were determined using a Dionex ICS-2100 ion chromatograph fitted with an AS11-HC column and AG11-HC guard column, EGC III KOH eluent generator, and ASRS 300 suppressor. Samples were neutralized using 0.1 M NaOH prior to analysis (relative standard deviation <5%) using a KOH gradient from 1 mM to 50 mM for optimal peak separation. Level of detection for acetate was 0.1 mg L−1.
For analysis of CH4 and CO2 concentrations and isotopic composition, porewater was immediately filtered to 0.7 μm in the field using Whatman glass-fiber filters, then stored in pre-evacuated glass vials sealed with butyl stoppers. Phosphoric acid was added to each sample to preserve for shipment to Florida State University (FSU). Samples were analyzed for CH4 and CO2 concentrations and stable isotopic composition (δ13C) on a ThermoFinnigan Delta-V Isotope Ratio Mass Spectrometer using the headspace equilibration method with He. Each sample was analyzed twice, and the average results for each sample were recorded. Analytical precision was 0.2‰ for 13C.
Preparation of Δ14C-DIC samples was done at FSU by He stripping and cryogenic purification, and the resultant pure CO2 was transferred to 6 mm tubes for Δ14C analysis at the National Ocean Sciences Accelerator Mass Spectrometry Facility. CO2 was prepared as graphite targets, and analyzed by accelerator mass spectrometry37. Values are reported according to the Δ notation put forth in ref. 38. The Δ notation normalizes the radiocarbon content of a sample to a nominal δ13C value (–25‰) and the collection time. The scale is linear and starts at –1000‰ when a sample has essentially 0% modern carbon, which would represent petroleum residue39. Analytical precision was 2‰ for 14C.
Methanogen abundance
Following the same protocol that was used throughout the DPH experimental phase22, soil cores were collected in parallel with CH4 and CO2 production measurements, 1–2 times per year during the growing season, from 20 to 30, 40 to 50, 75 to 100, 150 to 175-cm depth increments. Soil cores were immediately frozen on dry ice, shipped to the Georgia Institute of Technology (GT), and stored at −80 °C until analysis. The total DNA was extracted from homogenized peat samples using the DNeasy PowerSoil Kit (Qiagen, formerly MoBio PowerSoil DNA extraction kit) as previously described22,29. The abundance of mcrA gene was targeted to assess the methanogen population using primer pairs ME3F, ME2R40. All quantitative polymerase chain reaction (qPCR) assays were performed in triplicate on a StepOnePlus platform (Applied Biosystems StepOne Plus) with PowerUp SYBR Green Master Mix (Applied Biosystems StepOne Plus). To estimate the size of the methanogenic community, the tenfold serial dilution was constructed that ranged from 102 to 107 molecules of the standard. pGEM-T Easy plasmid (Promega) containing mcrA gene fragment from Methanococcus maripaludis S2 was used to obtain the standard curve. In addition, a no-template DNA control was run on each qPCR plate to exclude or detect any possible contamination. Finally, the melting curve and gel electrophoresis analyses were performed to confirm the specificity of the qPCR reaction. The abundance of mcrA gene copy numbers was calculated and presented as gene copy numbers per dry gram of peat.
Water-table position
Water-table absolute elevations (a.m.s.l.) were determined at the center of all instrumented enclosures using a TruTrack Water Height Probe (Model WT-VO 2000) suspended in a 5-cm diameter stainless-steel screen well (Driller Service, Inc.—DSI) installed to an approximate depth of 1.7 m. Surveyed well cap heights were the basis for water heights along the 2000-mm probe. These absolute elevations were further reconciled to enclosure-mean hollow heights to yield water-table levels above (positive) or below (negative) the mean hollow height. Cross-enclosure comparisons of hollow-relative water-table levels demanded further adjustments for calibration shifts using direct manual observations of the presence of surface water or measured subsurface water levels.
Surface CH4 and CO2 flux
A community-level flux measurement system41,42 was used to simultaneously measure CO2 and CH4 exchange at a spatial resolution that allowed for the inclusion of a representative sample of the aboveground community, including hummocks and hollows (with the exception of the widely spaced tree canopy). Briefly, we paired open-path sensors for the characterization of changing CO2 and CH4 concentrations within a sealed and nontransparent enclosure. A 1.2 m internal diameter flux collar was permanently placed in the bog embedded 10 to 20 cm into the surface peat to achieve an air-tight seal at ground level. Because of variable hummock-hollow topography, the volume of the collared portion of the enclosures ranged in volume from 0.45 to 0.69 m3.
Measurements of CH4 and CO2 emissions were made 6–8 times per year during 2015–2018. The transportable and nontransparent enclosure (1.25 m3) included fans for mixing, while no fans were located within the collar area to limit surface boundary layer disturbance. Total enclosed volume during measurements was around 1.8 m3. We used open-path CO2 × H2O (LI-7500A; LiCor Inc., Lincoln, NB) and CH4 (LI-7700) infrared sensors41,42.
Statistical analyses
General linear mixed-effect models were used to determine the effects of depth, temperature, and elevated CO2 concentrations on gas production (CH4 and CO2 production and CO2:CH4 ratios), porewater concentration (H2, acetate, CH4, and CO2 concentrations and CO2:CH4 ratios), microbial (methanogen abundance), and gas emission (CH4 and CO2 emissions and CO2:CH4 ratios) data sets. In all cases, enclosure was treated as a random effect, and all other predictor variables were analyzed as fixed effects. If significant differences among depths were detected (p < 0.05), pairwise comparisons using Tukey’s honest significant difference test (p < 0.05) were conducted. If not significantly different, depths were combined for linear regression analysis. In addition, stepwise multiple linear regression with Akaike Information Criterion (AIC) as the model selection condition was used to assess the ability of temperature, water-table position, and exposure to elevated CO2 conditions to predict peatland CH4 emissions. Data were tested for normality and log-transformed where the transformation resulted in an improvement in overall distribution. The above statistical analyses were completed using R 3.2.2 Statistical Software.
The LOESS (locally estimated scatterplot smoothing) regression for the radiocarbon plots was accomplished using a weighted least square and 2nd degree polynomial model via the smooth function in MATLAB 2017b (MathWorks, Inc).
Source: Ecology - nature.com


