Levine, J. M. & D’Antonio, C. M. Forecasting biological invasions with increasing international trade. Conserv. Biol. 17, 322–326 (2003).
Simberloff, D. et al. Impacts of biological invasions: what’s what and the way forward. Trends Ecol. Evol. 28, 58–66 (2013).
Pimentel, D. Biological invasions: economic and environmental costs of alien plant, animal, and microbe species. (CRC Press, 2014).
Lockwood, J. L., Hoopes, M. F. & Marchetti, M. P. Invasion ecology. (John Wiley & Sons, 2013).
Pimentel, D., Zuniga, R. & Morrison, D. Update on the environmental and economic costs associated with alien-invasive species in the United States. Ecol. Econ. 52, 273–288 (2005).
Pimentel, D., Lach, L., Zuniga, R. & Morrison, D. Environmental and economic costs of nonindigenous species in the United States. Bioscience 50, 53–66 (2000).
Vilà, M. & Hulme, P. E. Impact of biological invasions on ecosystem services. Vol. 12 (Springer, 2017).
Bucciarelli, G. M., Blaustein, A. R., Garcia, T. S. & Kats, L. B. Invasion complexities: the diverse impacts of nonnative species on amphibians. Copeia 2014, 611–632 (2014).
Nunes, A. L. et al. A global meta-analysis of the ecological impacts of alien species on native amphibians. Proc. R. Soc. Lond. [Biol.] 286, 20182528 (2019).
Gall, B. G. & Mathis, A. Innate predator recognition and the problem of introduced trout. Ethology 116, 47–58 (2010).
Bennett, A. M., Pereira, D. & Murray, D. L. Investment into defensive traits by anuran prey (Lithobates pipiens) is mediated by the starvation-predation risk trade-off. PLoS One 8, e82344 (2013).
Cadi, A. & Joly, P. Impact of the introduction of the red-eared slider (Trachemys scripta elegans) on survival rates of the European pond turtle (Emys orbicularis). Biodivers. Conserv. 13, 2511–2518 (2004).
Pearl, C. A., Adams, M. J., Schuytema, G. S. & Nebeker, A. V. Behavioral responses of anuran larvae to chemical cues of native and introduced predators in the Pacific Northwestern United States. J. Herpetol. 37, 572–577 (2003).
Lima, S. L. & Dill, L. M. Behavioral decisions made under the risk of predation: a review and prospectus. Can. J. Zool. 68, 619–640 (1990).
Petranka, J. W., Kats, L. B. & Sih, A. Predator-prey interactions among fish and larval amphibians: use of chemical cues to detect predatory fish. Anim. Behav. 35, 420–425 (1987).
Lefcort, H. & Eiger, S. M. Antipredatory behaviour of feverish tadpoles: implications for pathogen transmission. Behaviour 126, 13–27 (1993).
Kats, L. B. & Dill, L. M. The scent of death: chemosensory assessment of predation risk by prey animals. Ecoscience 5, 361–394 (1998).
Berec, M., Klapka, V. & Zemek, R. Effect of an alien turtle predator on movement activity of European brown frog tadpoles. Ital. J. Zool. 83, 68–76 (2016).
Lenda, M., Witek, M., Skórka, P., Moroń, D. & Woyciechowski, M. Invasive alien plants affect grassland ant communities, colony size and foraging behaviour. Biol. Invasions 15, 2403–2414 (2013).
Robbins, T. R., Freidenfelds, N. A. & Langkilde, T. Native predator eats invasive toxic prey: evidence for increased incidence of consumption rather than aversion-learning. Biol. Invasions 15, 407–415 (2013).
Márquez-García, A. Z., Campos-Verduzco, R. & Castro-Soriano, B. S. Sedimentología y morfología de la playa de anidación para tortugas marinas, El Carrizal, Coyuca de Benítez, Guerrero. Hidrobiológica 20, 101–112 (2010).
Gomez-Mestre, I. et al. The shape of things to come: linking developmental plasticity to post‐metamorphic morphology in anurans. J. Evol. Biol. 23, 1364–1373 (2010).
Goldberg, T., Nevo, E. & Degani, G. Phenotypic plasticity in larval development of six amphibian species in stressful natural environments. Zool. Stud. 51, 345–361 (2012).
Van Buskirk, J. The costs of an inducible defense in anuran larvae. Ecology 81, 2813–2821 (2000).
Bulen, B. J. & Distel, C. A. Carbaryl concentration gradients in realistic environments and their influence on our understanding of the tadpole food web. Arch. Env. Contam. Toxicol. 60, 343–350 (2011).
Nunes, A. et al. Antipredator responses of two anurans towards native and exotic predators. Amphibia-Reptilia 32, 341–350 (2011).
Phillips, B. L. & Shine, R. An invasive species induces rapid adaptive change in a native predator: cane toads and black snakes in Australia. Proc. R. Soc. Lond. [Biol.] 273, 1545–1550 (2006).
Berthon, K. How do native species respond to invaders? Mechanistic and trait-based perspectives. Biol. Invasions 17, 2199–2211 (2015).
Berchtold, A. Behavioural and physiological responses of prey fish to an invasive predator MSc thesis, Simon Fraser University, (2017).
Zhang, F., Zhao, J., Zhang, Y., Messenger, K. & Wang, Y. Antipredator behavioral responses of native and exotic tadpoles to novel predator. Asian Herpetol. Res. 6, 51–58 (2015).
Werner, E. E. Amphibian metamorphosis: growth rate, predation risk, and the optimal size at transformation. Am. Nat. 128, 319–341 (1986).
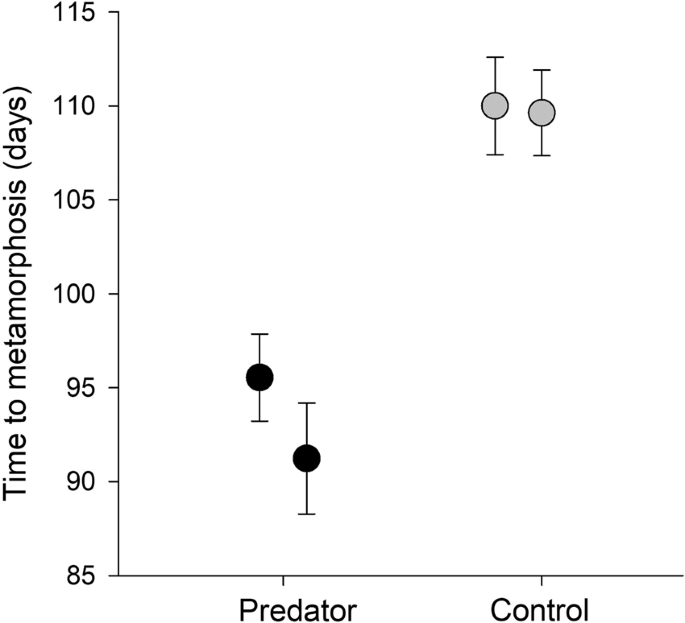
Relyea, R. A. Getting out alive: how predators affect the decision to metamorphose. Oecologia 152, 389–400 (2007).
Ernst, C. H. & Lovich, J. E. Turtles of the United States and Canada. (JHU Press, 2009).
Works, A. J. & Olson, D. H. Diets of two nonnative freshwater turtle species (Trachemys scripta and Pelodiscus sinensis) in Kawai Nui Marsh, Hawaii. J. Herpetol. 52, 444–452 (2018).
Ficetola, G. F., Rödder, D. & Padoa-Schioppa, E. In Handbook of global freshwater invasive species (ed. Francis, R.) 331–339 (Earthscan, Taylor and Francis Group, 2012).
Kraus, F. Alien reptiles and amphibians: a scientific compendium and analysis. Vol. 4 (Springer, 2009).
West-Eberhard, M. J. Phenotypic plasticity and the origins of diversity. Annu. Rev. Ecol. Syst. 20, 249–278 (1989).
Gosner, K. L. A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 16, 183–190 (1960).
Cox, J. G. & Lima, S. L. Naiveté and an aquatic–terrestrial dichotomy in the effects of introduced predators. Trends Ecol. Evol. 21, 674–680 (2006).
Banks, P. B. & Dickman, C. R. Alien predation and the effects of multiple levels of prey naiveté. Trends Ecol. Evol. 5, 229–230 (2007).
Hettyey, A. et al. Naive tadpoles do not recognize recent invasive predatory fishes as dangerous. Ecology 97, 2975–2985 (2016).
Sih, A. et al. Predator–prey naïveté, antipredator behavior, and the ecology of predator invasions. Oikos 119, 610–621 (2010).
Ferrari, M. C., Gonzalo, A., Messier, F. & Chivers, D. P. Generalization of learned predator recognition: an experimental test and framework for future studies. Proc. R. Soc. Lond. [Biol.] 274, 1853–1859 (2007).
Chalcraft, D. R. & Resetarits, W. J. Jr. Predator identity and ecological impacts: functional redundancy or functional diversity? Ecology 84, 2407–2418 (2003).
Sih, A., Englund, G. & Wooster, D. Emergent impacts of multiple predators on prey. Trends Ecol. Evol. 13, 350–355 (1998).
Soluk, D. A. & Collins, N. C. Synergistic interactions between fish and stoneflies: facilitation and interference among stream predators. Oikos 52, 94–100 (1988).
Stearns, S. C. The evolutionary significance of phenotypic plasticity. Bioscience 39, 436–445 (1989).
Newman, R. A. Adaptive plasticity in amphibian metamorphosis. Bioscience 42, 671–678 (1992).
Semlitsch, R. D. & Caldwell, J. P. Effects of density of growth, metamorphosis, and survivorship in tadpoles of Scaphiopus holbrooki. Ecology 63, 905–911 (1982).
Denver, R. J. Environmental stress as a developmental cue: corticotropin-releasing hormone is a proximate mediator of adaptive phenotypic plasticity in amphibian metamorphosis. Horm. Behav. 31, 169–179 (1997).
Wilbur, H. M. & Collins, J. P. Ecological aspects of amphibian metamorphosis: nonnormal distributions of competitive ability reflect selection for facultative metamorphosis. Science 182, 1305–1314 (1973).
Kiesecker, J. M., Chivers, D. P., Anderson, M. & Blaustein, A. R. Effect of predator diet on life history shifts of red-legged frogs, Rana aurora. J. Chem. Ecol. 28, 1007–1015 (2002).
Laurila, A., Kujasalo, J. & Ranta, E. Predator-induced changes in life history in two anuran tadpoles: effects of predator diet. Oikos 83, 307–317 (1998).
Kurzava, L. M. & Morin, P. J. Tests of functional equivalence: complementary roles of salamanders and fish in community organization. Ecology 79, 477–489 (1998).
Laurila, A. & Kujasalo, J. Habitat duration, predation risk and phenotypic plasticity in common frog (Rana temporaria) tadpoles. J. Anim. Ecol. 68, 1123–1132 (1999).
Boorse, G. C. & Denver, R. J. Acceleration of Ambystoma tigrinum metamorphosis by corticotropin‐releasing hormone. J. Exp. Zool. 293, 94–98 (2002).
Ruthsatz, K. et al. Endocrine disruption alters developmental energy allocation and performance in Rana temporaria. Integr. Comp. Biol. 59, 70–88 (2019).
Formanowicz, D. R. Jr. Anuran tadpole/aquatic insect predator-prey interactions: tadpole size and predator capture success. Herpetologica 42, 367–373 (1986).
Smith, D. C. Factors controlling tadpole populations of the chorus frog (Pseudacris triseriata) on Isle Royale, Michigan. Ecology 64, 501–510 (1983).
Bringsøe, H. In Handbuch der Reptilien und Amphibien Europas Vol. 3/IIIA Schildkröten (Testudines) I. (ed. Fritz, U.) 525–583 (Aula, 2001).
Source: Ecology - nature.com



