Ceballos, G. et al. Accelerated modern human–induced species losses: Entering the sixth mass extinction. Sci. Adv. 1, e1400253 (2015).
Secretariat of the Convention on Biological Diversity Global Biodiversity Outlook 4 (World Trade Centre, 2014).
Wright, S. Evolution in Mendelian populations. Genetics 16, 97–159 (1931).
Charlesworth, B. Effective population size and patterns of molecular evolution and variation. Nat. Rev. Genet. 10, 195–205 (2009).
Frankham, R. Effective population size/adult population size ratios in wildlife: a review. Genet. Res. 66, 95–107 (1995).
Leffler, E. M. et al. Revisiting an old riddle: what determines genetic diversity levels within species? PLoS Biol. 10, e1001388 (2012).
Lewontin, R. C. The Genetic Basis of Evolutionary Change (Columbia Univ. Press, 1974).
Ellegren, H. & Galtier, N. Determinants of genetic diversity. Nat. Rev. Genet. 17, 422–433 (2016).
Lynch, M. et al. Genetic drift, selection and the evolution of the mutation rate. Nat. Rev. Genet. 17, 704–714 (2016).
Cutter, A. D. & Payseur, B. A. Genomic signatures of selection at linked sites: unifying the disparity among species. Nat. Rev. Genet. 14, 262–274 (2013).
Corbett-Detig, R. B., Hartl, D. L. & Sackton, T. B. Natural selection constrains neutral diversity across a wide range of species. PLoS Biol. 13, e1002112 (2015).
Coop, G. Does linked selection explain the narrow range of genetic diversity across species? Preprint at bioRxiv https://doi.org/10.1101/042598 (2016).
Romiguier, J. et al. Comparative population genomics in animals uncovers the determinants of genetic diversity. Nature 515, 261–263 (2014).
Ferchaud, A.-L. et al. Making sense of the relationships between Ne, Nb and Nc towards defining conservation thresholds in atlantic salmon (Salmo salar). Heredity 117, 268–278 (2016).
Lande, R. Genetics and demography in biological conservation. Science 241, 1455–1460 (1988).
Palstra, F. P. & Fraser, D. J. Effective/census population size ratio estimation: a compendium and appraisal. Ecol. Evol. 2, 2357–2365 (2012).
Waples, R. S. Making sense of genetic estimates of effective population size. Mol. Ecol. 25, 4689–4691 (2016).
Palstra, F. P. & Ruzzante, D. E. Genetic estimates of contemporary effective population size: what can they tell us about the importance of genetic stochasticity for wild population persistence? Mol. Ecol. 17, 3428–3447 (2008).
Waples, R. S., Luikart, G., Faulkner, J. R. & Tallmon, D. A. Simple life-history traits explain key effective population size ratios across diverse taxa. Proc. R. Soc. B 280, 20131339 (2013).
Leroy, G. et al. Methods to estimate effective population size using pedigree data: examples in dog, sheep, cattle and horse. Genet. Sel. Evol. 45, 1 (2013).
Wang, J. Estimation of effective population sizes from data on genetic markers. Phil. Trans. R. Soc. B 360, 1395–1409 (2005).
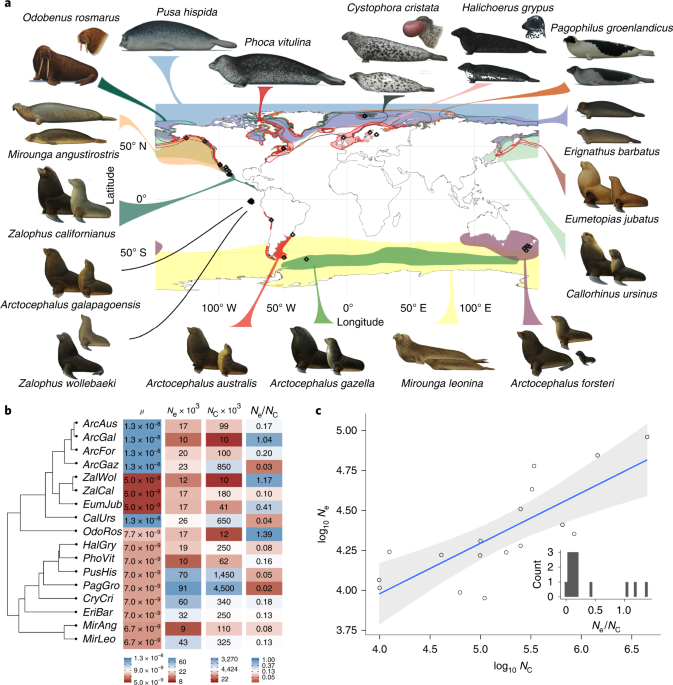
Stoffel, M. A. et al. Demographic histories and genetic diversity across pinnipeds are shaped by human exploitation, ecology and life-history. Nat. Commun. 9, 4836 (2018).
Tenesa, A. et al. Recent human effective population size estimated from linkage disequilibrium. Genome Res. 17, 520–526 (2007).
Nordborg, M. & Krone, S. M. in Modern Developments in Theoretical Population Genetics: The Legacy of Gustave Malécot (eds Slatkin, M. & Veuille, M.) 194–232 (Oxford Univ. Press, 2002).
Vijay, N. et al. Genomewide patterns of variation in genetic diversity are shared among populations, species and higher-order taxa. Mol. Ecol. 26, 4284–4295 (2017).
Wakeley, J. Coalescent Theory: An Introduction (W. H. Freeman, 2008).
Charlesworth, B. & Jain, K. Purifying selection, drift, and reversible mutation with arbitrarily high mutation rates. Genetics 198, 1587–1602 (2014).
Krüger, O., Wolf, J. B. W., Jonker, R. M., Hoffman, J. I. & Trillmich, F. Disentangling the contribution of sexual selection and ecology to the evolution of size dimorphism in pinnipeds. Evolution 68, 1485–1496 (2014).
de Oliveira, L. R., Meyer, D., Hoffman, J., Majluf, P. & Morgante, J. S. Evidence of a genetic bottleneck in an El Niño affected population of South American fur seals, Arctocephalus australis. J. Mar. Biol. Assoc. U.K. 89, 1717–1725 (2009).
Soto, K. H., Trites, A. W. & Arias-Schreiber, M. The effects of prey availability on pup mortality and the timing of birth of South American sea lions (Otaria flavescens) in Peru. J. Zool. 264, 419–428 (2004).
Kovacs, K. M. et al. Global threats to pinnipeds. Mar. Mammal Sci. 28, 414–436 (2012).
Scally, A. The mutation rate in human evolution and demographic inference. Curr. Opin. Genet. Dev. 41, 36–43 (2016).
Takahata, N. Allelic genealogy and human evolution. Mol. Biol. Evol. 10, 2–22 (1993).
Brüniche-Olsen, A. et al. The inference of gray whale (Eschrichtius robustus) historical population attributes from whole-genome sequences. BMC Evol. Biol. 18, 87 (2018).
Nei, M. & Takahata, N. Effective population size, genetic diversity, and coalescence time in subdivided populations. J. Mol. Evol. 37, 240–244 (1993).
Schiffels, S. & Durbin, R. Inferring human population size and separation history from multiple genome sequences. Nat. Genet. 46, 919 (2014).
Mazet, O., Rodríguez, W., Grusea, S., Boitard, S. & Chikhi, L. On the importance of being structured: instantaneous coalescence rates and human evolution—lessons for ancestral population size inference? Heredity 116, 362–371 (2016).
Andersen, L. W. et al. Walruses (Odobenus rosmarus rosmarus) in the Pechora Sea in the context of contemporary population structure of Northeast Atlantic walruses. Biol. J. Linn. Soc. 122, 897–915 (2017).
Kalinowski, S. T. & Waples, R. S. Relationship of effective to census size in fluctuating populations. Conserv. Biol. 16, 129–136 (2002).
Gutenkunst, R. N., Hernandez, R. D., Williamson, S. H. & Bustamante, C. D. Inferring the joint demographic history of multiple populations from multidimensional SNP frequency data. PLoS Genet. 5, e1000695 (2009).
Nyman, T. et al. Demographic histories and genetic diversities of Fennoscandian marine and landlocked ringed seal subspecies. Ecol. Evol. 4, 3420–3434 (2014).
Chikhi, L., Sousa, V. C., Luisi, P., Goossens, B. & Beaumont, M. A. The confounding effects of population structure, genetic diversity and the sampling scheme on the detection and quantification of population size changes. Genetics 186, 983–995 (2010).
Mackintosh, A. et al. The determinants of genetic diversity in butterflies. Nat. Commun. 10, 3466 (2019).
Wright, S. Size of population and breeding structure in relation to evolution. Science 87, 430–431 (1938).
Slatkin, M. Gene genealogies within mutant allelic classes. Genetics 143, 579–587 (1996).
Lancaster, M. L., Gemmell, N. J., Negro, S., Goldsworthy, S. & Sunnucks, P. Ménage à trois on Macquarie Island: hybridization among three species of fur seal (Arctocephalus spp.) following historical population extinction. Mol. Ecol. 15, 3681–3692 (2006).
Akcakaya, H. R. et al. Making consistent IUCN classifications under uncertainty. Conserv. Biol. 14, 1001–1013 (2000).
Higdon, J. W., Bininda-Emonds, O. R. P., Beck, R. M. D. & Ferguson, S. H. Phylogeny and divergence of the pinnipeds (Carnivora: Mammalia) assessed using a multigene dataset. BMC Evol. Biol. 7, 216 (2007).
de Oliveira, L. R. & Brownell, R. L. Taxonomic status of two subspecies of South American fur seals: Arctocephalus australis australis vs. A. a. gracilis. Mar. Mammal Sci. 30, 1258–1263 (2014).
Shafer, A. B. A. et al. Bioinformatic processing of RAD-seq data dramatically impacts downstream population genetic inference. Methods Ecol. Evol. 8, 907–917 (2017).
Brelsford, A., Dufresnes, C. & Perrin, N. High-density sex-specific linkage maps of a European tree frog (Hyla arborea) identify the sex chromosome without information on offspring sex. Heredity 116, 177–181 (2016).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 17, 10 (2011).
Catchen, J., Hohenlohe, P. A., Bassham, S., Amores, A. & Cresko, W. A. Stacks: an analysis tool set for population genomics. Mol. Ecol. 22, 3124–3140 (2013).
Zhang, J., Kobert, K., Flouri, T. & Stamatakis, A. PEAR: a fast and accurate Illumina Paired-End reAd mergeR. Bioinformatics 30, 614–620 (2014).
Nadeau, N. J. et al. Population genomics of parallel hybrid zones in the mimetic butterflies, H. melpomene and H. erato. Genome Res. 24, 1316–1333 (2014).
Foote, A. D. et al. Convergent evolution of the genomes of marine mammals. Nat. Genet. 47, 272–275 (2015).
Humble, E. et al. RAD sequencing and a hybrid Antarctic fur seal genome assembly reveal rapidly decaying linkage disequilibrium, global population structure and evidence for inbreeding. G3 (Bethesda) 8, 2709–2722 (2018).
Lunter, G. & Goodson, M. Stampy: a statistical algorithm for sensitive and fast mapping of Illumina sequence reads. Genome Res. 21, 936–939 (2011).
Korneliussen, T. S., Albrechtsen, A. & Nielsen, R. ANGSD: analysis of next generation sequencing data. BMC Bioinform. 15, 356 (2014).
Harris, R. S. Improved Pairwise Alignment of Genomic DNA (The Pennsylvania State Univ., 2007).
Fumagalli, M., Vieira, F. G., Linderoth, T. & Nielsen, R. ngsTools: methods for population genetics analyses from next-generation sequencing data. Bioinformatics 30, 1486–1487 (2014).
Fumagalli, M. et al. Quantifying population genetic differentiation from next-generation sequencing data. Genetics 195, 979–992 (2013).
Nei, M. & Li, W. H. Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc. Natl Acad. Sci. USA 76, 5269–5273 (1979).
Watterson, G. A. On the number of segregating sites in genetical models without recombination. Theor. Popul. Biol. 7, 256–276 (1975).
Tajima, F. Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123, 585–595 (1989).
Hudson, R. R., Slatkin, M. & Maddison, W. P. Estimation of levels of gene flow from DNA sequence data. Genetics 132, 583 (1992).
Ranwez, V. et al. OrthoMaM: a database of orthologous genomic markers for placental mammal phylogenetics. BMC Evol. Biol. 7, 241 (2007).
Ranwez, V., Harispe, S., Delsuc, F. & Douzery, E. J. P. MACSE: multiple alignment of coding sequences accounting for frameshifts and stop codons. PLoS ONE 6, e22594 (2011).
Di Franco, A., Poujol, R., Baurain, D. & Philippe, H. Evaluating the usefulness of alignment filtering methods to reduce the impact of errors on evolutionary inferences. BMC Evol. Biol. 19, 21 (2019).
Romiguier, J. et al. Fast and robust characterization of time-heterogeneous sequence evolutionary processes using substitution mapping. PLoS ONE 7, e33852 (2012).
Guéguen, L. et al. Bio++: efficient extensible libraries and tools for computational molecular evolution. Mol. Biol. Evol. 30, 1745–1750 (2013).
Figuet, E. et al. Life history traits, protein evolution, and the nearly neutral theory in amniotes. Mol. Biol. Evol. 33, 1517–1527 (2016).
Botero-Castro, F., Figuet, E., Tilak, M.-K., Nabholz, B. & Galtier, N. Avian genomes revisited: hidden genes uncovered and the rates versus traits paradox in birds. Mol. Biol. Evol. 34, 3123–3131 (2017).
The IUCN Red List of Threatened Species. Version 2017-3 (IUCN, 2017).
Shafer, A. B. A., Gattepaille, L. M., Stewart, R. E. A. & Wolf, J. B. W. Demographic inferences using short-read genomic data in an approximate Bayesian computation framework: in silico evaluation of power, biases and proof of concept in Atlantic walrus. Mol. Ecol. 24, 328–345 (2015).
Warmuth, V. M. & Ellegren, H. Genotype‐free estimation of allele frequencies reduces bias and improves demographic inference from RADSeq data. Mol. Ecol. Resour. 19, 586–596 (2019).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Li, H. et al. The sequence alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Bearded Seal (Greenland Institute of Natural Resources, 2018); http://www.natur.gl/en/birds-and-mammals/marine-mammals/bearded-seal/
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2018).
Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D. & R Core Team. nlme: Linear and nonlinear mixed effects models. R package version 3.1 (2017).
Source: Ecology - nature.com



