Li, W., Podar, M. & Morgan-Kiss, R. M. Ultrastructural and Single-Cell-Level Characterization Reveals Metabolic Versatility in a Microbial Eukaryote Community from an Ice-Covered Antarctic Lake. Appl Environ Microbiol. 82, 3659–3670, https://doi.org/10.1128/AEM.00478-16 (2016).
Fokin, S. I. Bacterial endocytobionts of ciliophora and their interactions with the host cell. Int Rev Cytol. 236, 181–249, https://doi.org/10.1016/S0074-7696(04)36005-5 (2004).
Gortz, H. D. Intracellular bacteria in ciliates. Int Microbiol. 4, 143–150, https://doi.org/10.1007/s10123-001-0029-9 (2001).
Petroni, G., Spring, S., Schleifer, K. H., Verni, F. & Rosati, G. Defensive extrusive ectosymbionts of Euplotidium (Ciliophora) that contain microtubule-like structures are bacteria related to Verrucomicrobia. Proc Natl Acad Sci USA. 97, 1813–1817, https://doi.org/10.1073/pnas.030438197 (2000).
Gast, R. J., Moran, D. M., Dennett, M. R. & Caron, D. A. Kleptoplasty in an Antarctic dinoflagellate: caught in evolutionary transition? Environ Microbiol. 9, 39–45, https://doi.org/10.1111/j.1462-2920.2006.01109.x (2007).
Schrallhammer, M., Schweikert, M., Vallesi, A., Verni, F. & Petroni, G. Detection of a novel subspecies of Francisella noatunensis as endosymbiont of the ciliate Euplotes raikovi. Microb Ecol. 61, 455–464, https://doi.org/10.1007/s00248-010-9772-9 (2011).
Valbonesi, A. & Luporini, P. Biology of Euplotes focardii, an Antarctic ciliate. Polar Biol. 13, 489–493 (1993).
La Terza, A., Papa, G., Miceli, C. & Luporini, P. Divergence between two Antarctic species of the ciliate Euplotes, E. focardii and E. nobilii, in the expression of heat-shock protein 70 genes. Mol Ecol. 10, 1061–1067 (2001).
Pucciarelli, S. et al. Molecular cold-adaptation of protein function and gene regulation: The case for comparative genomic analyses in marine ciliated protozoa. Mar. Genomics. 2, 57–66, https://doi.org/10.1016/j.margen.2009.03.008 (2009).
Chiappori, F. et al. Structural thermal adaptation of beta-tubulins from the Antarctic psychrophilic protozoan Euplotes focardii. Proteins. 80, 1154–1166, https://doi.org/10.1002/prot.24016 (2012).
Marziale, F. et al. Different roles of two gamma-tubulin isotypes in the cytoskeleton of the Antarctic ciliate Euplotes focardii: remodelling of interaction surfaces may enhance microtubule nucleation at low temperature. FEBS J. 275, 5367–5382, https://doi.org/10.1111/j.1742-4658.2008.06666.x (2008).
Pucciarelli, S. et al. Tubulin folding: the special case of a beta-tubulin isotype from the Antarctic psychrophilic ciliate Euplotes focardii. Polar Biology. 36, 1833–1838, https://doi.org/10.1007/s00300-013-1390-9 (2013).
Pucciarelli, S. & Miceli, C. Characterization of the cold-adapted alpha-tubulin from the psychrophilic ciliate Euplotes focardii. Extremophiles. 6, 385–389, https://doi.org/10.1007/s00792-002-0268-5 (2002).
Pischedda, A. et al. Antarctic marine ciliates under stress: superoxide dismutases from the psychrophilic Euplotes focardii are cold-active yet heat tolerant enzymes. Sci Rep. 8, 14721, https://doi.org/10.1038/s41598-018-33127-1 (2018).
Yang, G. et al. Characterization of the first eukaryotic cold-adapted patatin-like phospholipase from the psychrophilic Euplotes focardii: Identification of putative determinants of thermal-adaptation by comparison with the homologous protein from the mesophilic Euplotes crassus. Biochimie. 95, 1795–1806, https://doi.org/10.1016/j.biochi.2013.06.008 (2013).
Yang, G. et al. Characterization and comparative analysis of psychrophilic and mesophilic alpha-amylases from Euplotes species: a contribution to the understanding of enzyme thermal adaptation. Biochem Biophys Res Commun. 438, 715–720, https://doi.org/10.1016/j.bbrc.2013.07.113 (2013).
Yang, G. et al. Rational engineering of a cold-adapted alpha-amylase from the Antarctic ciliate Euplotes focardii for simultaneous improvement of thermostability and catalytic activity. Appl Environ Microbiol., https://doi.org/10.1128/AEM.00449-17 (2017).
Lobanov, A. V. et al. Position-dependent termination and widespread obligatory frameshifting in Euplotes translation. Nat Struct Mol Biol. 24, 61–68, https://doi.org/10.1038/nsmb.3330 (2017).
Pucciarelli, S. et al. Microbial Consortium Associated with the Antarctic Marine Ciliate Euplotes focardii: An Investigation from Genomic Sequences. Microb Ecol. 70, 484–497, https://doi.org/10.1007/s00248-015-0568-9 (2015).
Juhas, M. et al. Genomic islands: tools of bacterial horizontal gene transfer and evolution. FEMS Microbiol Rev. 33, 376–393, https://doi.org/10.1111/j.1574-6976.2008.00136.x (2009).
Soucy, S. M., Huang, J. & Gogarten, J. P. Horizontal gene transfer: building the web of life. Nat Rev Genet. 16, 472–482, https://doi.org/10.1038/nrg3962 (2015).
Baumann, L., Baumann, P., Mandel, M. & Allen, R. D. Taxonomy of aerobic marine eubacteria. J Bacteriol. 110, 402–429 (1972).
Kajikazawa, T., Sugita, T. & Nishikawa, A. Comprehensive identification of bacteria in processed fresh edible sea urchin using 16S ribosomal DNA sequence analysis: the products contain various food poisoning-related bacteria and opportunistic bacterial pathogens. Journal of Health Science. 53, 756–759 (2007).
Prabagaran, S. R., Suresh, K., Manorama, R. & Delille, D. & Shivaji, S. Marinomonas ushuaiensis sp. nov., isolated from coastal sea water in Ushuaia, Argentina, sub-Antarctica. Int J Syst Evol Microbiol. 55, 309–313, https://doi.org/10.1099/ijs.0.63363-0 (2005).
Mangiagalli, M. et al. The co‐existence of cold activity and thermal stability in an Antarctic GH42 β‐galactosidase relies on its hexameric quaternary arrangement. FEBS J. https://doi.org/10.1111/febs.15354 (2020).
Iravani, S., Korbekandi, H., Mirmohammadi, S. V. & Zolfaghari, B. Synthesis of silver nanoparticles: chemical, physical and biological methods. Res Pharm Sci. 9, 385–406 (2014).
Gurunathan, S. et al. Biosynthesis, purification and characterization of silver nanoparticles using Escherichia coli. Colloids Surf B Biointerfaces. 74, 328–335, https://doi.org/10.1016/j.colsurfb.2009.07.048 (2009).
Silver, S., Phung le, T. & Silver, G. Silver as biocides in burn and wound dressings and bacterial resistance to silver compounds. J Ind Microbiol Biotechnol. 33, 627–634, https://doi.org/10.1007/s10295-006-0139-7 (2006).
Fujikawa, H. & Morozumi, S. Modeling surface growth of Escherichia coli on agar plates. Appl Environ Microbiol. 71, 7920–7926, https://doi.org/10.1128/AEM.71.12.7920-7926.2005 (2005).
Jukes, T. H. & Cantor, C. R. In Mammalian Protein Metabolism (ed. Munro, H. N.) 21–132 (Academic Press, New York, 1969).
Jones, D. T., Taylor, W. R. & Thornton, J. M. The rapid generation of mutation data matrices from protein sequences. Comput Appl Biosci. 8, 275–282 (1992).
Tamura, K. et al. MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol. 28, 2731–2739, https://doi.org/10.1093/molbev/msr121 (2011).
Magoč, T., & Salzberg, S. L. FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics (Oxford, England), 27(21), 2957–2963. https://doi.org/10.1093/bioinformatics/btr507 (2011).
Margulies, M. et al. Genome sequencing in microfabricated high-density picolitre reactors. Nature. 437, 376–380, https://doi.org/10.1038/nature03959 (2005).
Seemann, T. Prokka: rapid prokaryotic genome annotation. Bioinformatics. 30, 2068–2069, https://doi.org/10.1093/bioinformatics/btu153 (2014).
Gotz, S. et al. High-throughput functional annotation and data mining with the Blast2GO suite. Nucleic Acids Res. 36, 3420–3435, https://doi.org/10.1093/nar/gkn176 (2008).
Birolo, G. & Telatin, A. covtobed: a simple and fast tool to extract coverage tracks from BAM files. Journal of Open Source Software, 5(47), 2119, https://doi.org/10.21105/joss.02119 (2020).
Page, M., MacLean, D., & Schudoma, C. blastjs: a BLAST+ wrapper for Node.js. BMC research notes, 9, 130. https://doi.org/10.1186/s13104-016-1938-1 (2016).
Darling, A. C., Mau, B., Blattner, F. R. & Perna, N. T. Mauve: multiple alignment of conserved genomic sequence with rearrangements. Genome Res. 14, 1394–1403, https://doi.org/10.1101/gr.2289704 (2004).
Benjamini, Y. & Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. Journal of the Royal Statistical Society. Series B (Methodological). 57, 289–300 (1995).
Natalello, A. et al. Co-fibrillogenesis of Wild-type and D76N beta2-Microglobulin: The Crucial Role of Fibrillar Seeds. J Biol Chem. 291, 9678–9689, https://doi.org/10.1074/jbc.M116.720573 (2016).
Bauer, A. W., Kirby, W. M., Sherris, J. C. & Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol. 45, 493–496 (1966).
CLSI. in CLSI document EP100-S23 (Wayne, P.A.: Clinical and Laboratory Standards Institute, 2019).
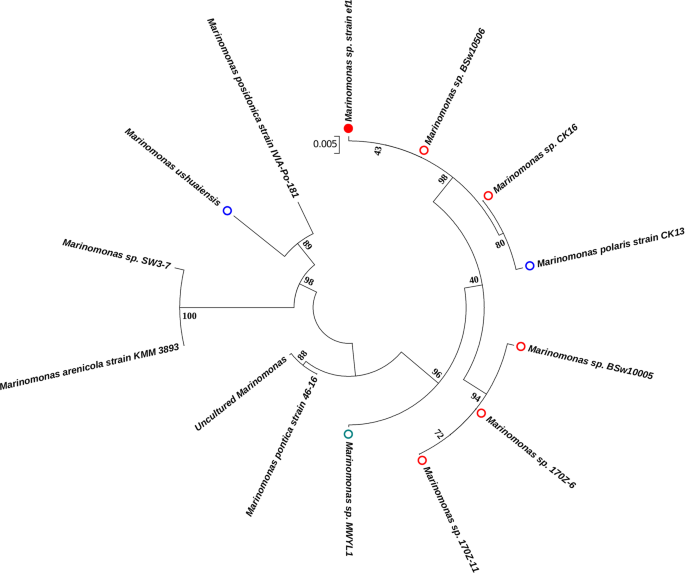
Ramasamy, K. P., Telatin, A., Mozzicafreddo, M., Miceli, C. & Pucciarelli, S. Draft Genome Sequence of a New Pseudomonas sp. Strain, ef1, Associated with the Psychrophilic Antarctic Ciliate Euplotes focardii. Microbiol Resour Announc. 8, https://doi.org/10.1128/MRA.00867-19 (2019).
John, M. S. et al. Synthesis of Bioactive Silver Nanoparticles by a Pseudomonas Strain Associated with the Antarctic Psychrophilic Protozoon Euplotes focardii. Mar Drugs. 18, https://doi.org/10.3390/md18010038 (2020).
Zhang, D. C. et al. Marinomonas arctica sp. nov., a psychrotolerant bacterium isolated from the Arctic. Int J Syst Evol Microbiol. 58, 1715–1718, https://doi.org/10.1099/ijs.0.65737-0 (2008).
Kotakadi, V. S., Gaddam, S. A., Venkata, S. K., Sarma, P. V. & Sai Gopal, D. V. Biofabrication and spectral characterization of silver nanoparticles and their cytotoxic studies on human CD34 +ve stem cells. 3 Biotech. 6, 216, https://doi.org/10.1007/s13205-016-0532-5 (2016).
Suresh, A. K. et al. Monodispersed biocompatible silver sulfide nanoparticles: facile extracellular biosynthesis using the gamma-proteobacterium, Shewanella oneidensis. Acta Biomater. 7, 4253–4258, https://doi.org/10.1016/j.actbio.2011.07.007 (2011).
Barth, A. Infrared spectroscopy of proteins. Biochimica et Biophysica Acta (BBA) – Bioenergetics. 1767, 1073–1101.
Baker, M. J. et al. Developing and understanding biofluid vibrational spectroscopy: a critical review. Chem Soc Rev. 45, 1803–1818, https://doi.org/10.1039/c5cs00585j (2016).
Naumann, D. In Encyclopedia of Analytical Chemistry (ed Meyers, R. A.) 102–131 (John Wiley & Sons, Ltd., 2000).
Barnes, D. K. & Clarke, A. Antarctic marine biology. Curr Biol. 21, R451–457, https://doi.org/10.1016/j.cub.2011.04.012 (2011).
Tian, R. M. et al. Genome Reduction and Microbe-Host Interactions Drive Adaptation of a Sulfur-Oxidizing Bacterium Associated with a Cold Seep Sponge. mSystems. 2, https://doi.org/10.1128/mSystems.00184-16 (2017).
Gottlieb, Y., Lalzar, I. & Klasson, L. Distinctive Genome Reduction Rates Revealed by Genomic Analyses of Two Coxiella-Like Endosymbionts in Ticks. Genome Biol Evol. 7, 1779–1796, https://doi.org/10.1093/gbe/evv108 (2015).
Baltrus, D. A. et al. Absence of genome reduction in diverse, facultative endohyphal bacteria. Microb Genom. 3, e000101, https://doi.org/10.1099/mgen.0.000101 (2017).
Koonin, E. V. & Wolf, Y. I. Genomics of bacteria and archaea: the emerging dynamic view of the prokaryotic world. Nucleic Acids Res. 36, 6688–6719, https://doi.org/10.1093/nar/gkn668 (2008).
Das, V. L. et al. Extracellular synthesis of silver nanoparticles by the Bacillus strain CS 11 isolated from industrialized area. 3 Biotech. 4, 121–126, https://doi.org/10.1007/s13205-013-0130-8 (2014).
Dipak, P. & Sankar, N. S. Extracellular synthesis of silver nanoparticles using pseudomonas sp KUPSB12 and its antibacterial activity. Jordan Journal of Biological Sciences., 245–250 (2014).
Thamilselvi, V. & Radha, K. V. Synthesis of silver nanoparticles from Pseudomonas putida ncim 2650 in silver nitrate supplemented growth medium and optimization using response surface methodology. Digest journal of nanomaterials and biostructures. 8, 1101–1111 (2013).
Lin, Y. S. E., Vidic, R. D., Stout, J. E., McCartney, C. A. & Yu, V. L. Inactivation of Mycobacterium avium by copper and silver ions. Water Research. 32, 1997–2000 (1998).
Rai, M., Yadav, A. & Gade, A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol Adv. 27, 76–83, https://doi.org/10.1016/j.biotechadv.2008.09.002 (2009).
Eckhardt, S. et al. Nanobio silver: its interactions with peptides and bacteria, and its uses in medicine. Chem Rev. 113, 4708–4754, https://doi.org/10.1021/cr300288v (2013).
Mancini, A., La Vigna, G., Puciarelli, S., Lombardi, F. E. & Barocci, S. A three-year study entailing molecular characterization and epidemiology of Clostridium difficile in an Italian tertiary care hospital. Infez Med. 26, 204–209 (2018).
Mancini, A. & Pucciarelli, S. Antibiotic activity of the antioxidant drink effective Microorganism-X (EM-X) extracts against common nosocomial pathogens: an in vitro study. Nat Prod Res., 1–6, https://doi.org/10.1080/14786419.2018.1517344 (2018).
Source: Ecology - nature.com



